📚 New Blog Posts
2. The Reality of Lot Traceability in Process Manufacturing
3. KPIs That Drive Enterprise Value in $10–$50M Process Manufacturers (Part 2 of 2)
A monthly update for process manufacturing leaders across food, pharmaceutical, nutraceutical, chemical, and other industries
🎯 EXECUTIVE BRIEF
What Process Manufacturing Leaders Need to Know This Month
If you operate a $5M–$50M process manufacturing business, this February 2026 edition highlights the buyer activity you’ve been waiting for. Manufacturing PMI hit 52.6%—the first expansion after 12 months of contraction—and private equity firms are deploying capital aggressively across food, pharma, chemical, nutraceutical, and personal care sectors.
Here’s what matters for your business: Operational documentation is now driving 30-50% valuation premiums. Recent specialty adhesives deal commanded 6.2x EBITDA (6.2 times annual earnings) versus the typical 4.5x—the difference? Complete batch records, deviation tracking, and audit-ready traceability systems.
Buyers aren’t chasing growth projections anymore. They’re paying premiums for what they can verify: repeatable processes, regulatory compliance, and integration-readiness. Whether you’re evaluating a sale in the next 12-24 months or building long-term value, operational maturity is your highest-ROI investment.
📊 BY THE NUMBERS
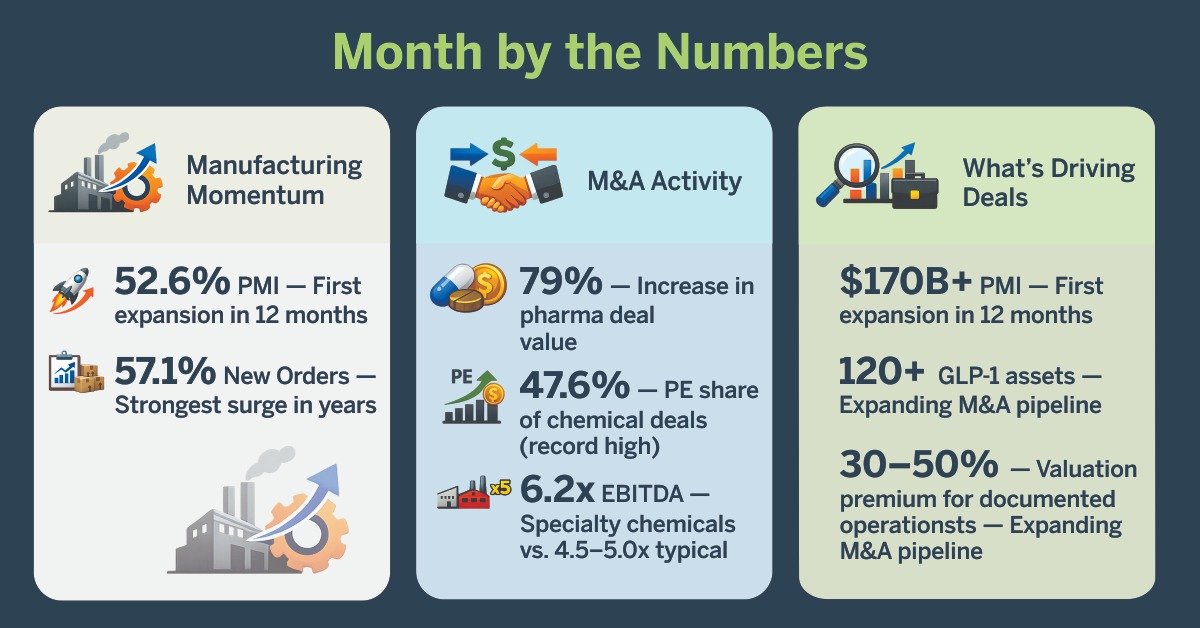
Manufacturing Expansion Returns:
- 52.6% — ISM Manufacturing Purchasing Managers’ Index (PMI) for January 2026, first expansion in 12 months and highest since February 2022
- 57.1% — New Orders Index surged 9.7 points, strongest reading in nearly four years
M&A Market Momentum:
- 79% — Pharma deal value increase, signaling decisive return of confidence
- 47.6% — Private equity share of chemical sector deals, highest on record
- 6.2x EBITDA — Recent specialty adhesives multiple (6.2 times annual earnings) vs. 4.5-5.0x typical range, driven by operational documentation
Sector-Specific Signals:
- $170B+ — Pharmaceutical patent cliff revenue at risk by 2032, driving aggressive M&A
- 120+ metabolic assets — GLP-1 obesity market creating deep M&A pipeline across 60 companies
- 30-50% — Valuation premium for businesses with documented operational excellence vs. those lacking process controls
🏭 PROCESS MANUFACTURING INSIGHTS
Pharmaceutical & Life Sciences Manufacturing
The $170B+ patent cliff looming by 2032 drives unprecedented acquisition urgency. Key trends:
- GLP-1 obesity market competition intensified with multiple bidding wars
- 80%+ of recent radio pharma deals included manufacturing integration—buyers want to own capacity, not just molecules
Buyer Focus: Late-stage clinical assets, next-generation delivery platforms, vertical integration, manufacturing capacity control.
Chemical Manufacturing (Specialty & Coatings)
January opened strong with key platform and add-on activity:
- Windjammer Capital acquired MFG Chemical from Platte River Equity (January 6)
- Riverside Company invested in Key Polymer as add-on to platform Seatex
- Integration-ready businesses commanding 5.5-6.5x EBITDA (5.5-6.5 times annual earnings)
- Water treatment businesses with 95%+ customer retention remain hot targets
- BASF’s AgBiTech acquisition (H1 2026 close) creates demand for qualified mid-market contract manufacturers
Buyer Focus: Repeatable formulations, operational discipline (>95% OTIF, <5% deviation rates), integration readiness, complete FDA/EPA documentation.
Nutraceutical & Supplement Manufacturing
Contract manufacturing consolidation accelerates as PE firms view CDMOs as defensive play. Key trends:
- 41% of consumers now trust AI recommendations for supplement guidance
- FDA proposed rule: single DSHEA disclaimer on labels (welcomed by industry)
- Functional wellness and practitioner-sold supplements with recurring revenue attract investment
- Precision nutrition and personalized supplement strategies gaining prominence
Buyer Focus: Contract manufacturers with vertical integration, science-backed formulations with third-party validation, professional channel recurring revenue models.
Food & Beverage Manufacturing
Better-for-you products and functional beverages continue attracting buyers:
- Manufacturing PMI strength signals improved demand outlook
- PE focuses on platform builds and bolt-on acquisitions with operational synergies
- Supply-chain excellence and tech-enabled operations top investment priorities
- Value-conscious consumers driving private label M&A growth
Buyer Focus: Brands with repeat-purchase economics, scalable margins, clean-label positioning, resilient demand fundamentals.
Personal Care & Cosmetics Manufacturing
Strategic buyers dominate with disciplined activity:
- Science-led clinical skincare and founder-led indie brands commanding premium valuations
- Hair care expected to accelerate significantly in 2026
- Contract manufacturing (CDMO/CMO) consolidation driven by nearshoring demands
- K-Beauty consolidation continues with cross-border deals
Buyer Focus: Founder-led brands, science-backed formulations with clinical validation, U.S. manufacturing capabilities, digital-native brands.
💸 NOTABLE DEALS
Chemical Manufacturing
1. Windjammer Capital Acquires MFG Chemical from Platte River Equity
Deal Type: PE buyout | Vertical: Specialty water treatment & coatings | Announced: January 6, 2026
Transaction exemplifies how repeatable formulations, flexible manufacturing, and defensible customer bases drive PE interest in mid-market specialty chemicals. MFG’s 95%+ customer retention and 98.5% OTIF delivery were critical value drivers.
Leadership Takeaways:
- CEOs/Owners: Customer retention metrics outweigh raw revenue growth. Build “why we win” narrative around operational reliability and relationship stickiness.
- CFOs/Controllers: Product-level profitability and customer concentration analysis are essential diligence deliverables. Prepare a detailed margin bridge.
- COOs: Batch record integrity and <5% deviation rate command premium multiples. Document QC consistency and delivery performance metrics.
- CTOs/IT: Audit-ready traceability systems accelerate diligence and reduce buyer risk perception. ERP compatibility matters for integration planning.
2. Riverside Company Invests in Key Polymer (Seatex Platform Add-On)
Deal Type: Add-on acquisition | Vertical: Adhesives & sealants | Announced: January 2026
Platform + add-on strategy demonstrates integration readiness, which drives deal execution. Key Polymer’s standardized processes and compatible ERP enabled rapid integration planning and premium valuation.
Leadership Takeaways:
- CEOs/Owners: Integration feasibility is as important as standalone value. Clean operational processes create platform buyer appeal.
- CFOs: Product-level profitability transparency enables synergy modeling and cross-selling identification during diligence.
- COOs: Process standardization and documentation quality impact integration timeline projections—key drivers of add-on valuations.
- IT/Systems: ERP compatibility with platform infrastructure reduces technical integration risk and accelerates close.
3. BASF to Acquire AgBiTech
Deal Type: Strategic expansion | Vertical: Agricultural biologicals | Expected Close: H1 2026
Large chemical companies expanding into biologics create immediate downstream demand for qualified contract manufacturers with regulatory-grade capabilities. Mid-market suppliers should evaluate second-source qualification opportunities.
Leadership Takeaways:
- CEOs: Strategic M&A by large players creates contract manufacturing opportunities. Position as a qualified second-source supplier.
- CFOs: Long-term supply agreements with strategic buyer’s command 20-30% premium pricing for regulatory-compliant production.
- COOs: Regulatory-grade intermediate production requires full traceability, environmental controls, FDA/EPA-compliant batch documentation.
- Quality/Regulatory: Second-source qualification audits are extensive. Proactive compliance documentation creates a competitive advantage.
Pharmaceutical & Life Sciences
Merck Acquires Cidara Therapeutics
Deal Value: ~$9.2B | Vertical: Respiratory/infectious disease | Announced: Late 2025
Merck pivots toward respiratory pipeline expansion by acquiring Cidara’s late-stage flu antiviral candidate. Deal demonstrates pharmaceutical urgency to fill patent cliffs with near-commercial assets.
Key Insight: Late-stage assets with clear commercial pathways for large addressable markets justify aggressive pricing.
Nutraceutical & Supplement
1. Market Trend: AI-Driven Supplement Discovery Accelerates
Development: January 2026
Consumer research shows 41% now trust AI recommendations for supplement guidance, shifting from traditional research methods. This raises stakes for accuracy, transparency, and brand education.
Leadership Implications: Transparency around ingredient sourcing and scientific validation matters more than ever. Brands fail to substantiate claims of risk misrepresentation in AI-driven recommendations.
2. FDA Proposes DSHEA Disclaimer Rule Change
Regulatory Update: January 2026
FDA proposed rule requiring only one DSHEA disclaimer on supplement labels welcomed by industry stakeholders. Change formalizes current practice but could forestall class action lawsuits.
Implication: Regulatory streamlining creates a favorable compliance environment. Focus shifts to substantiation and transparency.
3. FSMA Food Traceability Rule Compliance
FSMA Food Traceability Rule compliance deadline extended to July 2028; manufacturers must prepare for end-to-end supply chain traceability and electronic recordkeeping systems.
⚙️ WHAT BUYERS ARE REALLY PAYING FOR
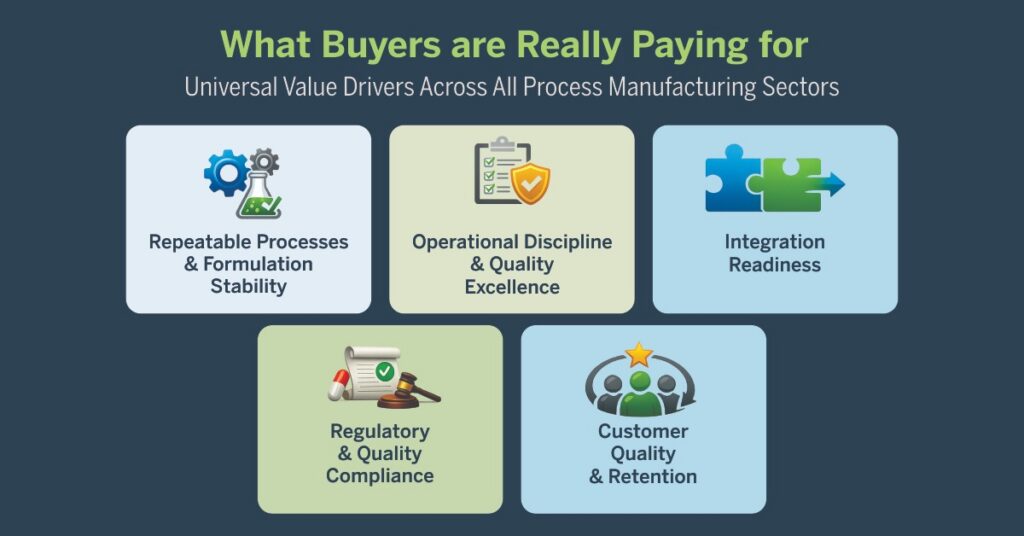
Universal Value Drivers Across All Process Manufacturing Sectors
1. Repeatable Processes & Formulation Stability
- Documented batch-to-batch consistency (<2% variation over 24+ months)
- Statistical process control and multi-year stability data
- Command 25-40% valuation premiums
- Recent example: 6.2x EBITDA (6.2 times annual earnings) vs. 4.5-5.0x typical range
2. Operational Discipline & Quality Excellence
- Complete batch records (24-36 months documentation)
- OTIF delivery >95%, deviation rates <5%
- Documented corrective actions
- Increases multiples by 1.0-1.5x across all verticals
3. Integration Readiness
- Clean data systems, standardized processes
- Transparent operations
- Close 20-30% faster, often receive exclusive negotiating periods
4. Regulatory & Quality Compliance
- Food: FDA compliance, GFSI certification, allergen control, traceability
- Pharma: cGMP documentation, FDA inspection history, quality system maturity
- Nutraceuticals: NSF/GMP certifications, third-party testing, substantiation data
- Chemicals: EPA permits, TSCA compliance, EHS documentation
- Personal Care: Cosmetic GMP, safety assessments, international compliance
5. Customer Quality & Retention
- Long-term relationships, retention rates >90%
- Documented quality scorecards, low complaint rates
- Create defensible competitive positions
- Example: MFG Chemical’s 95%+ retention cited as critical value driver
🎤 INDUSTRY VOICE
Operational Documentation: The 40% Valuation Premium
A California specialty adhesives manufacturer with $18M revenue completed a competitive auction in January that revealed how operational documentation drives premium valuations.
The company had solid margins (~45%) and steady growth (~8% CAGR). Differentiation came from comprehensive documentation: 36 months of complete batch records, detailed deviation tracking with root cause analysis, audit-ready traceability, and quality metrics showing <2% batch variation.
During three days of on-site diligence, the buyer reviewed processes and documentation. The competing bid—a business with 20% higher revenue—couldn’t produce complete batch records for 24 months.
“The buyer walked away from $22M in revenue because they couldn’t verify process consistency,” the owner explained. “They told us: ‘We can’t pay for revenue we can’t trust.'”
The California manufacturer commanded 6.2x EBITDA (6.2 times annual earnings)—40% above the 4.5x segment average—and closed in 87 days vs. 120–150-day typical timeline.
The lesson applies across all process verticals: Documented operational excellence drives 30-50% valuation premiums. Buyers reward businesses where they can verify—not just believe—that processes are repeatable, compliant, and integration-ready.
Critical documentation that drives premium valuations:
- 24-36 months complete batch production records (no gaps)
- Deviation logs with root cause analysis, corrective actions, CAPA closure
- Quality control data showing batch-to-batch consistency
- Customer quality scorecards, complaint logs, resolution tracking
- Environmental permits, safety certifications, training records, audit history
- Traceability from raw material receipt through finished goods shipment
- Maintenance logs, equipment calibration records, preventive maintenance schedules
Timeline: 18-24 months of consistent execution to create the track record buyers value most. Start now considering M&A in 12-24 months.
🔧 LEADERSHIP PLAYBOOK
What Leadership Should Focus on Now
For Finance Leaders (CFOs/Controllers)
- Build comprehensive margin bridge: product-level profitability, customer contribution, COGS drivers
- Document customer concentration: retention metrics, churn analysis, lifetime value
- Track KPIs: EBITDA margin trends, working capital efficiency, cash conversion cycle
- Prepare normalized financial statements with documented add-backs
For Operations Leaders (COOs/VP Operations)
- Audit batch records: ensure 24-36 months complete documentation with no gaps
- Review deviation management: root cause analysis, CAPA closure, trend analysis
- Optimize OTIF delivery: target >95% with weekly tracking
- Document processes: SOPs, process flows, control points, training records
- Implement statistical process control showing batch-to-batch consistency
For Technology & IT Leaders (CTOs/CIOs)
- Ensure traceability systems provide audit-ready reports from raw materials through finished goods
- Verify formulation control: version control, change management, access security
- Document system architecture, data dictionaries, integration points
- Review cybersecurity: network security, backup/recovery, disaster recovery plans
For R&D, Quality & Regulatory Leaders
- Food: Document formulations, allergen controls, nutritional claims, shelf-life studies
- Pharma: Maintain drug master files, method validation, stability programs, tech transfer
- Nutraceuticals: Third-party testing, COA archives, supplier qualifications
- Chemicals: Formulation IP protection, safety data sheets, regulatory compliance
- Personal Care: Safety assessments, stability studies, preservative efficacy, claim substantiation
- Maintain current registrations, licenses, permits with documented renewal dates
- Document regulatory inspection history, responses, CAPA closure evidence
Focus: Start now if you consider M&A in 12-24 months. Building operational documentation takes 18-24 months of consistent execution.
📜 POLICY & COMPLIANCE
Federal Regulatory Activity
FDA:
- Pre Check Program streamlines pharma oversight (increases documentation requirements)
- Advanced manufacturing guidance finalized
- Remote regulatory assessment guidance released
- FSMA emphasis on preventive controls and traceability continues
- DSHEA disclaimer rule proposed (single label disclaimer)
EPA:
- PFAS reporting rule imposes detailed requirements (even for unintentional presence)
- TSCA updates ongoing
- Emission compliance scrutiny increasing
Administration Developments:
- Most Favored Nations pricing policy affecting pharma dealmaking
- UK agreed to 25% drug price increases for 3-year tariff-free exports
- U.S. manufacturing investments exempt from additional tariffs
Sector-Specific Compliance
- Pharmaceuticals: Manufacturing capacity control becoming strategic priority; vertical integration accelerating
- Nutraceuticals: AI-driven discovery raising stakes for transparency; third-party certification now competitive requirement
- Chemicals: TSCA risk evaluations continuing; ag/biologicals expansion creating demand for regulatory-grade production
- Food & Beverage: FSMA Food Traceability Rule compliance deadline extended to July 2028; manufacturers must prepare for end-to-end supply chain traceability and electronic recordkeeping systems
Implication: Buyers discount incomplete regulatory documentation by 15-25%; proactive compliance drives premium valuations
💡 ONE-MINUTE EXECUTIVE TALKING POINTS
- Manufacturing rebound: ISM PMI 52.6% (first expansion in 12 months), New Orders 57.1%, capacity 82.4%
- Pharma M&A surge: 79% deal value increase—decisive confidence return
- Patent cliff urgency: $170B+ pharma revenue at risk by 2032; 120+ GLP-1 assets create deep pipeline
- Operational documentation premium: 30-50% valuation advantage—example: 6.2x EBITDA (6.2 times annual earnings) vs. 4.5x
- Chemical momentum: Windjammer/MFG and Riverside/Key Polymer demonstrate platform + add-on strength
- Vertical integration focus: Pharma buyers want manufacturing capacity, not just molecules—80%+ of deals included manufacturing
- Nutra AI shift: 41% trust AI for supplement guidance—raises transparency stakes
- Compliance = value: Incomplete documentation creates 15-25% discounts
- Timeline critical: 18-24 months needed to build documentation track record
- Cross-sector lesson: Operational maturity beats growth projections in 2026 M&A market
📞 CONNECT WITH US
Have insights to share? Email us at: news@batchmaster.com
Want to discuss how this impacts your business? We’re seeking operational leaders to share perspectives on manufacturing excellence and market trends.
📚 SOURCES & REFERENCES
Economic & Manufacturing Data:
- ISM Manufacturing PMI Report – January 2026
- 2025 Fourth Quarter Manufacturers’ Outlook Survey
- U.S. Federal Reserve – Industrial Production
M&A & Industry Intelligence:
- CNBC – Big Pharma Patent Cliff ($170B+)
- Pharmaceutical Technology – Biopharma 2025 M&A Boom
- HealthEconomics.com – Patent Cliffs Drive 2025 M&A
- RD WorldOnline – Life Sciences M&A Hit $240B
- Fierce Pharma – 2026 M&A Forecast
- Pharma Salmanac – How Pharma M&A Evolved in 2025
- DeepCeutix – $300B Revenue Loses Patent Protection
- Manufacturing Dive – PMI Expands in January
- Shutdown Deal Restricts Funds for FSMA 204, Produce Safety Rule Enforcement
Regulatory & Compliance:
- U.S. FDA – Newsroom
- U.S. EPA – Newsreleases
- American Chemistry Council
- American Herbal Products Association
Nutraceutical & Supplement Intelligence:
- New Hope Network
- Nutritional Outlook
- SPINS
- DataM Intelligence
Industry Associations:
- SOCMA (Society of Chemical Manufacturers & Affiliates)
- AIChE (American Institute of Chemical Engineers)
- Natural Products Association
- Personal Care Products Council
This Month in Process Manufacturing is published monthly by BatchMaster Software. It provides process manufacturing leaders with curated insights on M&A activity, regulatory trends, and operational best practices across food, pharmaceutical, nutraceutical, chemical, and personal care industries.

