Take Control of Pharmaceutical Manufacturing with an Industry-Specific ERP
BatchMaster Pharma ERP Software is an industry-specific ERP designed for the pharmaceutical industry. It brings complete efficiency across the supply chain, from regulatory compliance and formulation to quality control, production, and finished product.


- Ensure accurate potency calculations, theoretical yield tracking, and controlled batch scaling.
- Track materials by lot numbers, shelf-life, and expiry dates while leveraging FEFO logic, cold chain monitoring, and barcode integration for streamlined warehouse management.
- Stay compliant with global regulations like FDA 21 CFR Part 11 and cGMP through enforced SOP-based operations, audit trails, and electronic signatures.
- Enforce quality checks at every stage with stability testing, COA generation, and release workflows.
- Manage electronic manufacturing Batch Record (MBR) and BMR with built-in templates, real-time data capture, and version control.
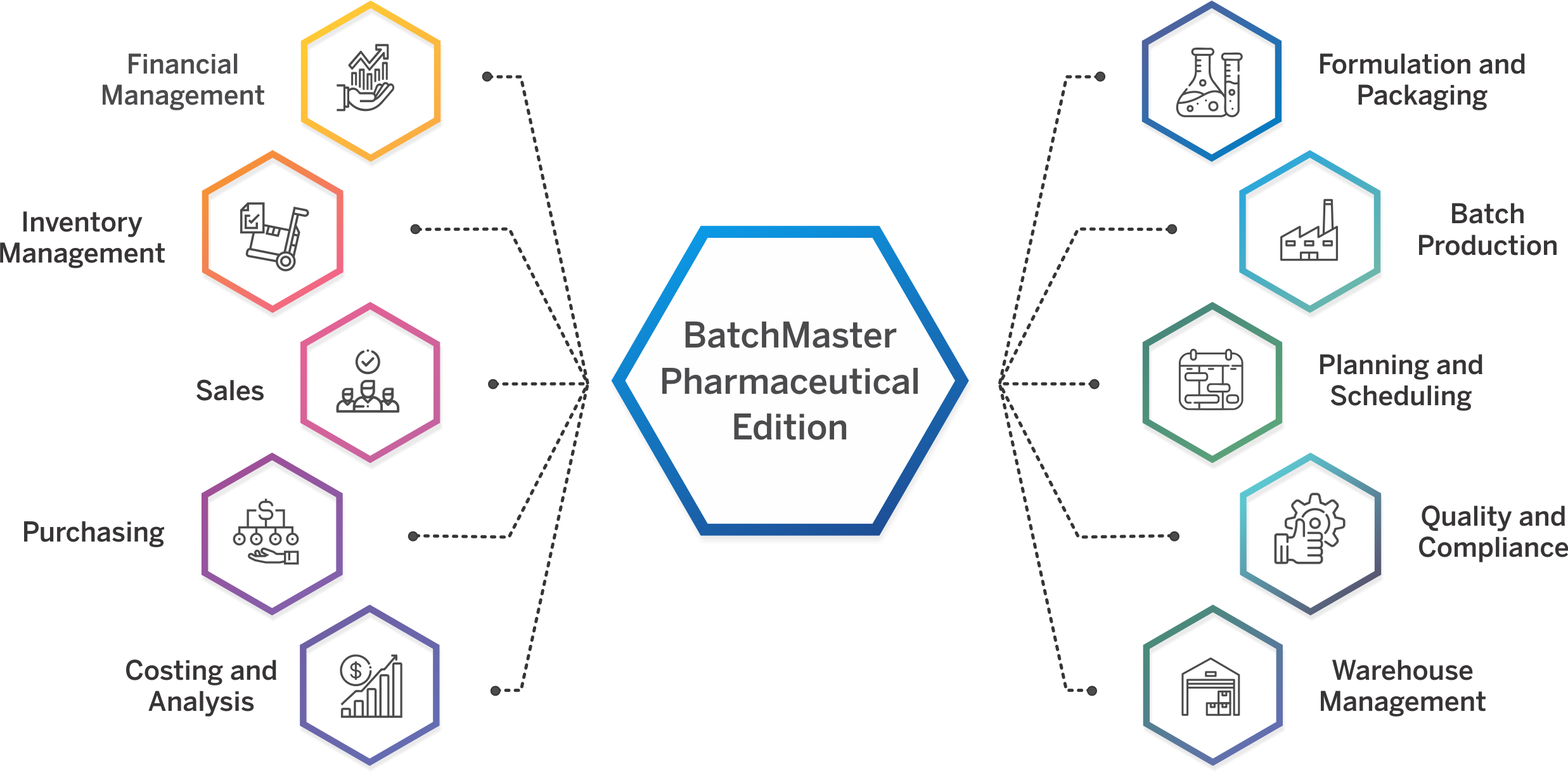
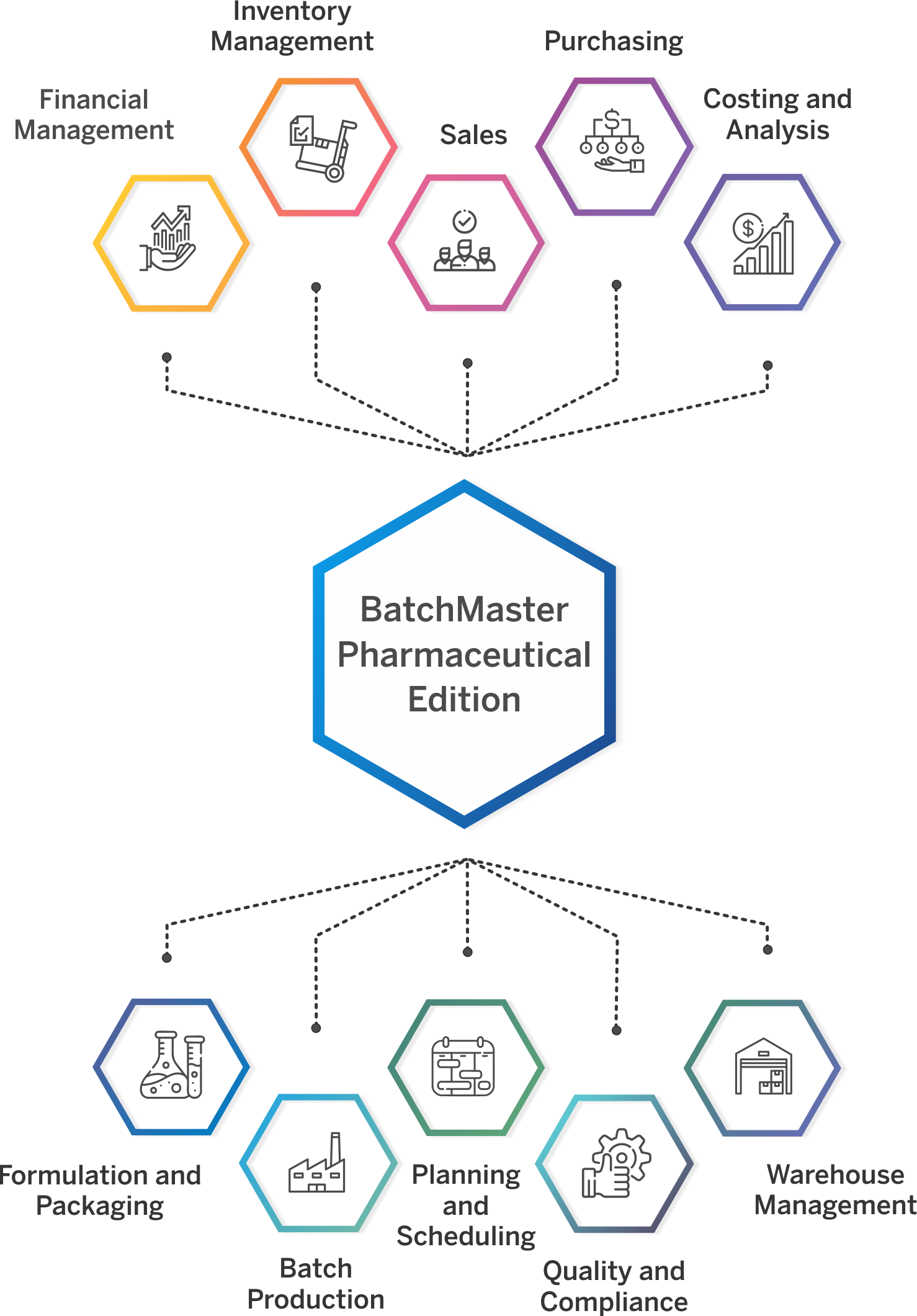
Pharmaceutical ERP Software That Meets Industry-Specific Needs
A modern ERP for pharma industry addresses complex pharmaceutical manufacturing requirements while unifying inventory, production, distribution, and financial capabilities in a single system.
Trusted by Leading Pharmaceutical Manufacturers
DRIVING SUCCESS ACROSS ALL PHARMACEUTICAL SEGMENTS
BatchMaster Pharma ERP Software caters to diverse segments through specialized pharmaceutical ERP modules designed to meet the unique requirements of each micro-vertical.

Active Pharmaceutical Ingredients

Biotechnology

Vaccines

Veterinary

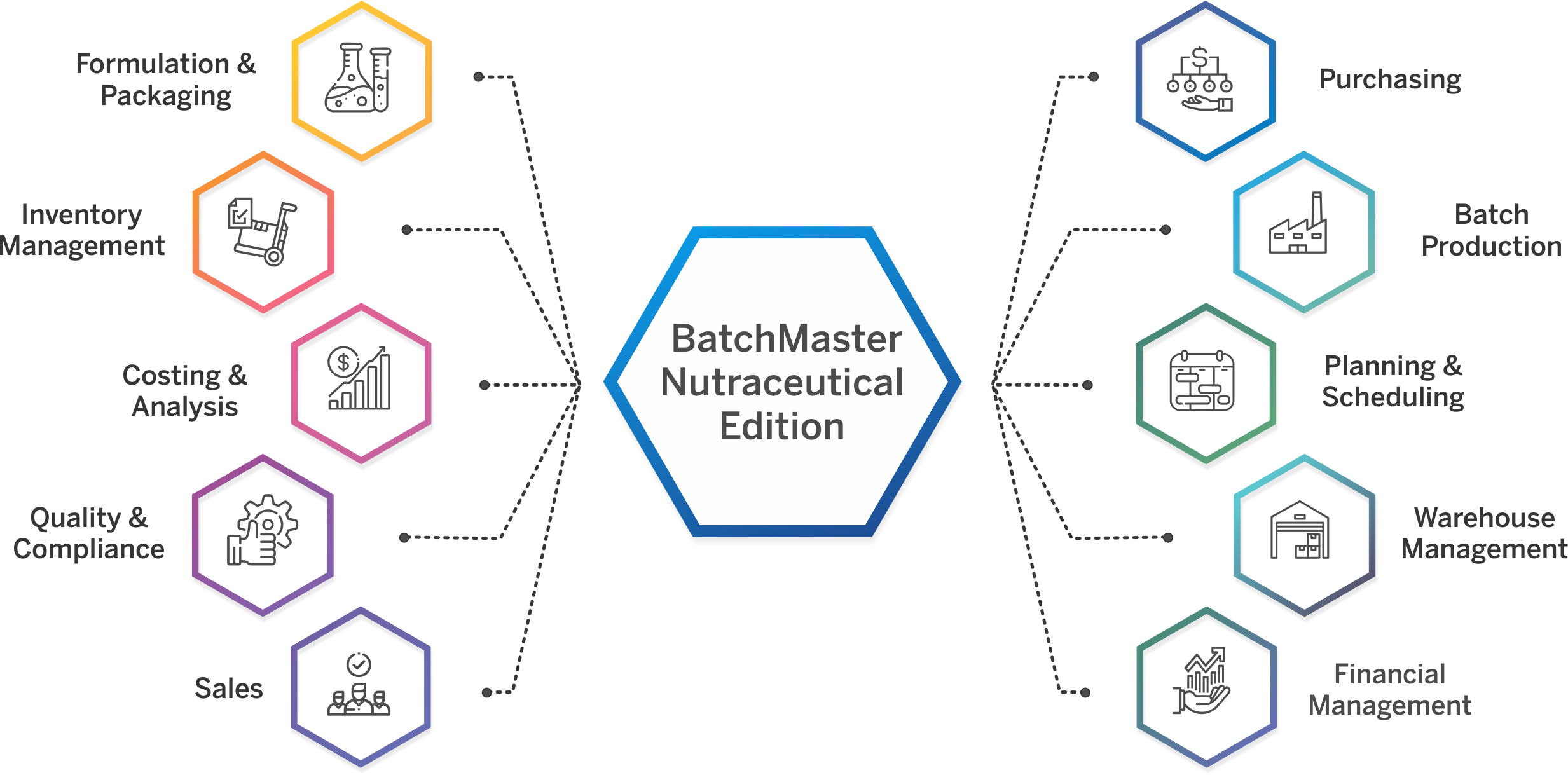
Nutraceuticals
Specialized Capabilities for Pharmaceutical Operations
Manage materials by active potency rather than just weight or volume. Automatically adjust input quantities in real-time to meet dosage requirements and ensure compliance with strength specifications.
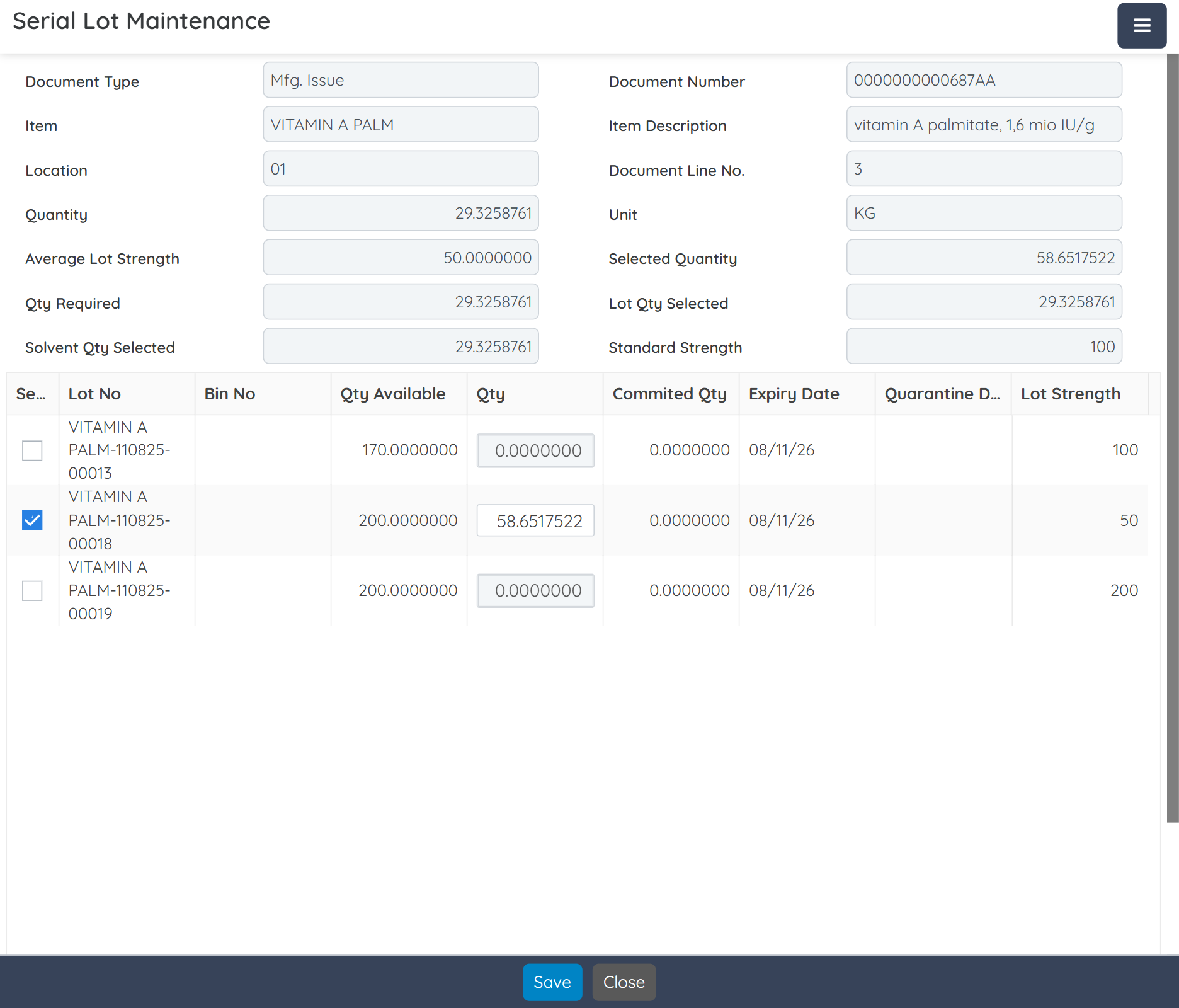
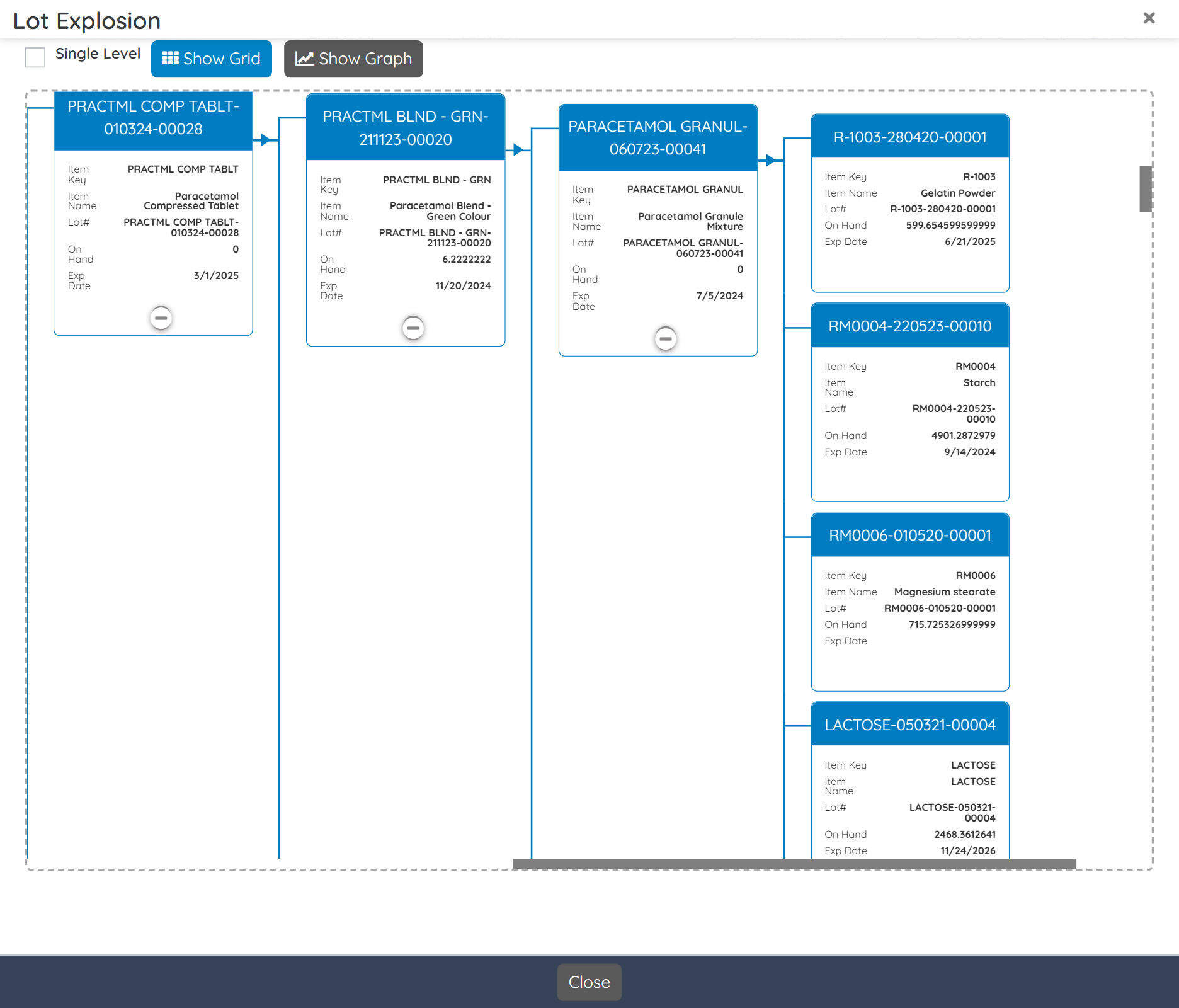
Digitally record every batch, from formulation and material consumption to yield and quality checks, eliminating errors and ensuring audit-ready traceability.
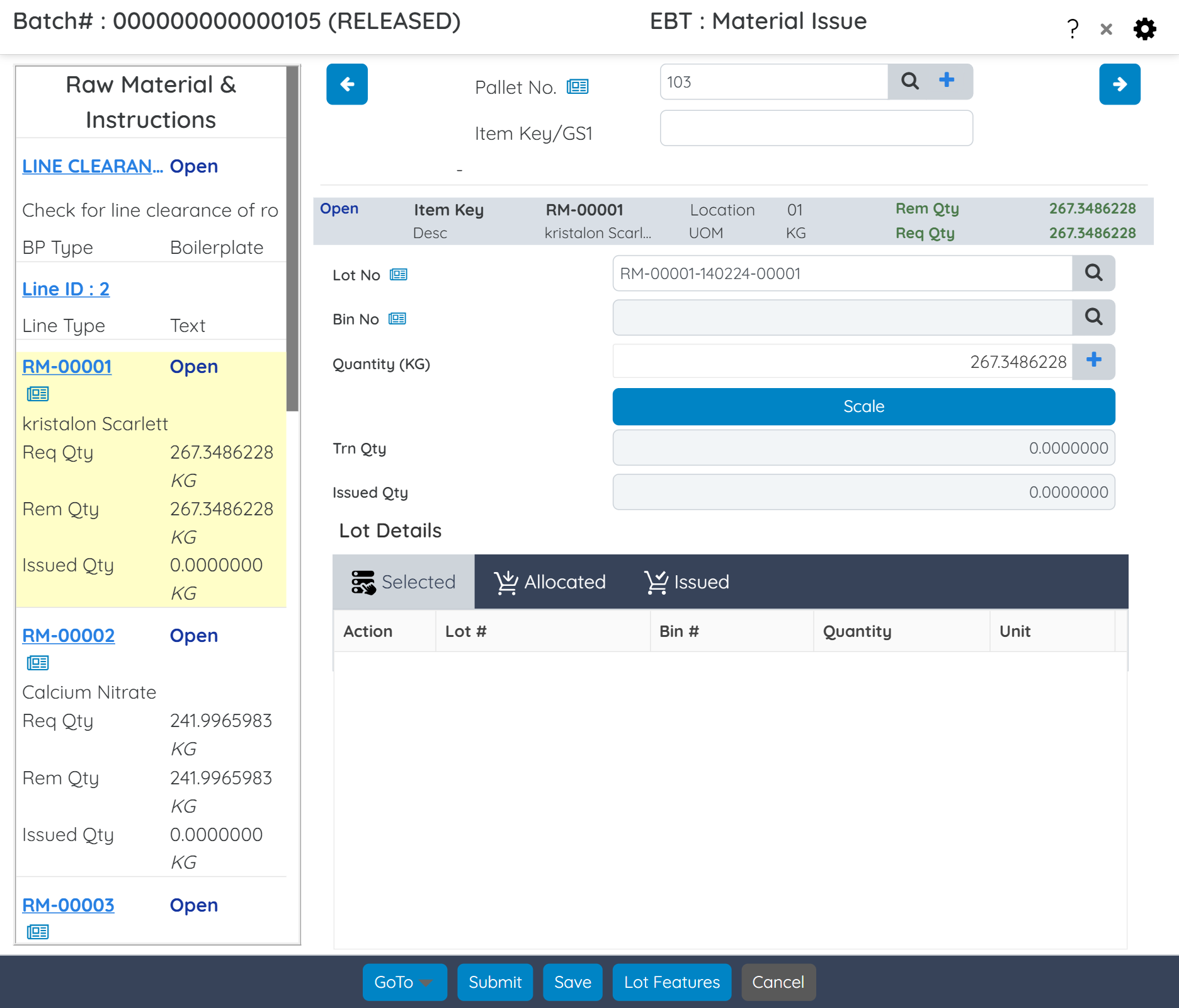
Enforce compliance with US FDA and global regulations through electronic signatures, secure access controls, and detailed audit trails across the manufacturing process, from material issue to batch release.
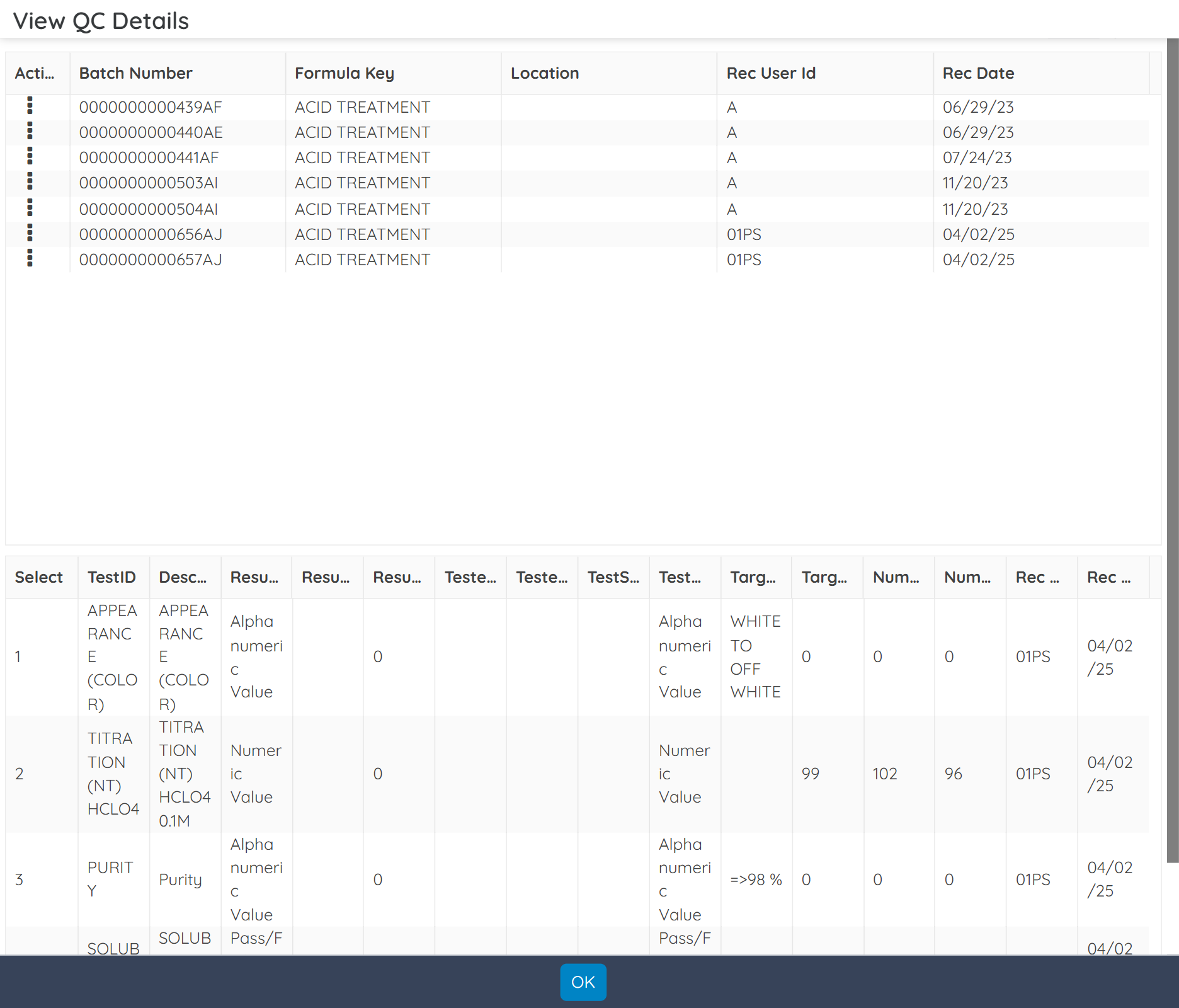
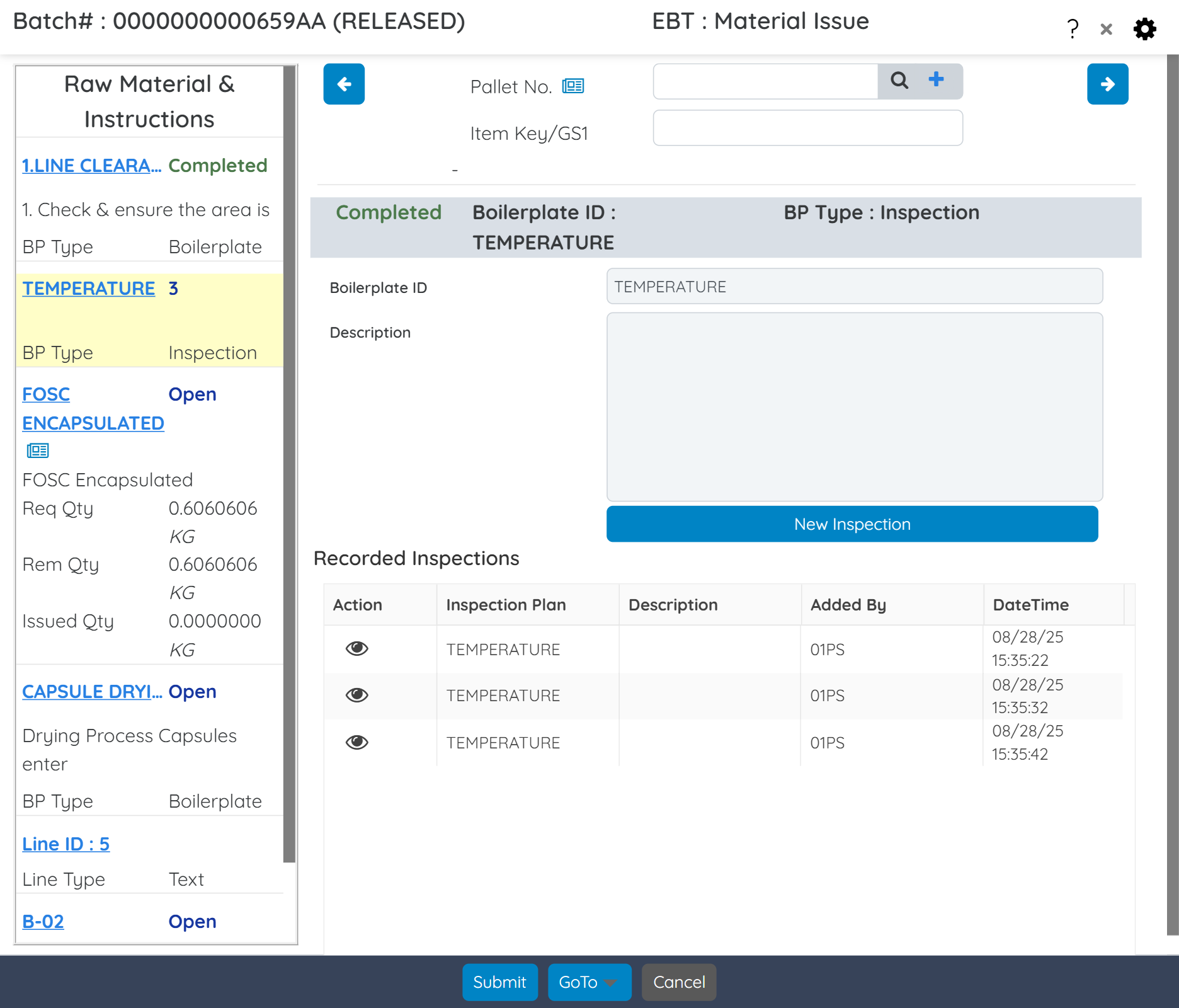
Schedule in process, stability, and shelf-life testing for raw materials and finished products. Define test parameters, record and analyze results, manage retesting schedules, and automate pass/fail workflows to ensure consistent quality.
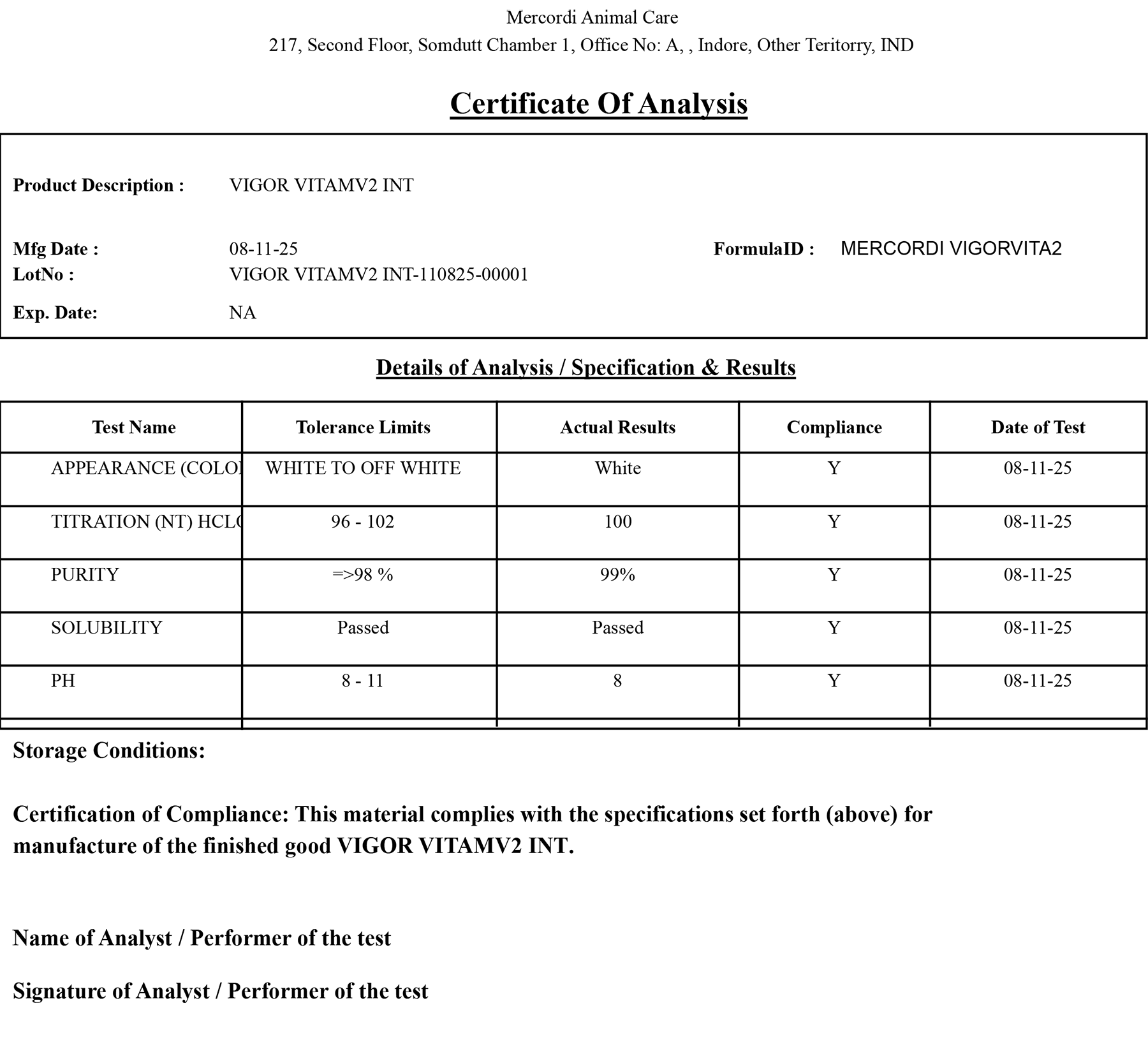
Automatically generate COAs at batch completion or during dispatch, integrating QC test results to meet customer and regulatory requirements with seamless documentation.
Manage and track inventory with FEFO logic, shelf-life monitoring, and expiration alerts. Get real-time visibility with barcode scanners, RF devices, mobile WMS, and electronic scales.
Streamline product development from R&D trails and batches to commercialization with version-controlled formulations, test results, and cost vs yield performance comparisons.
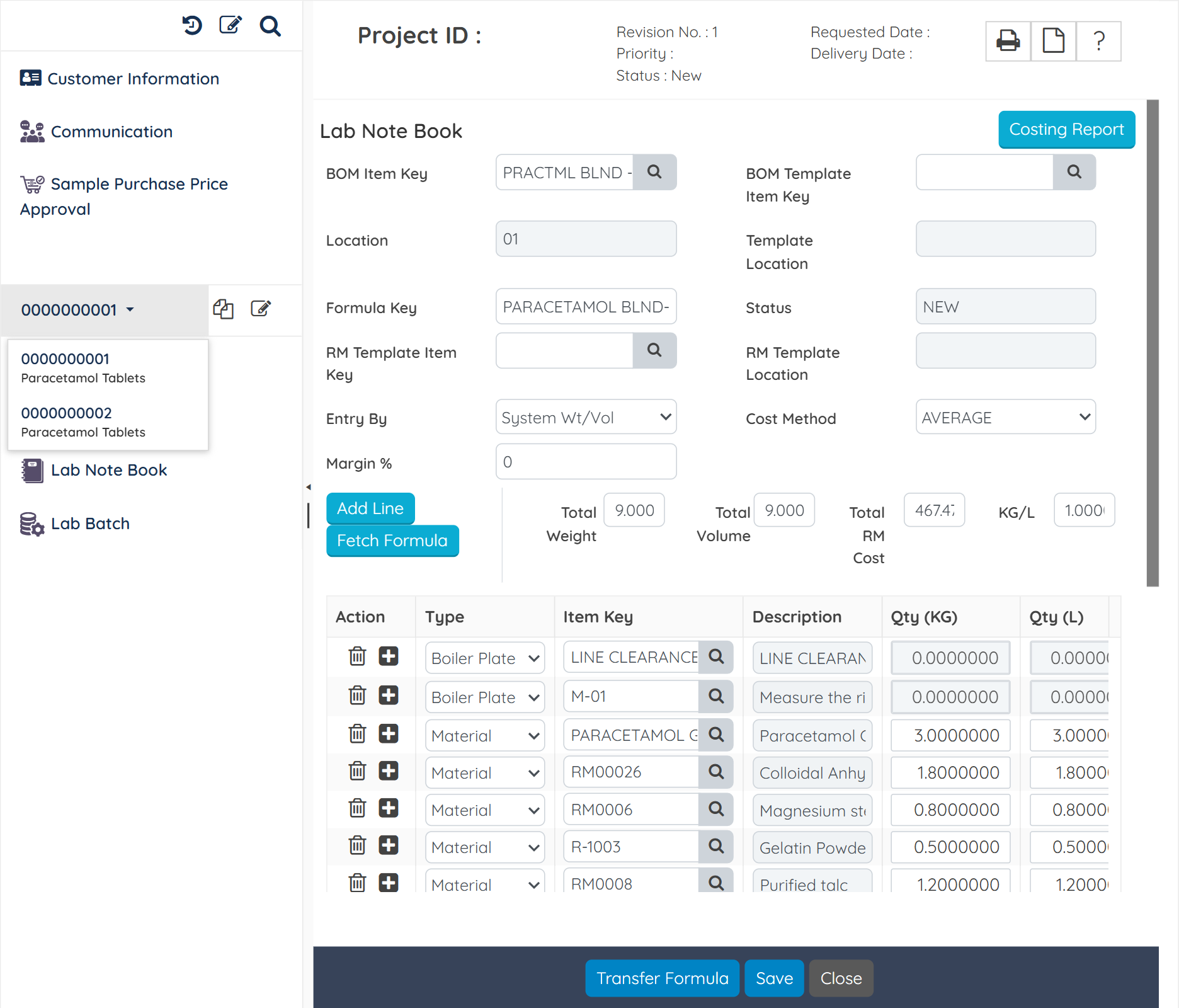
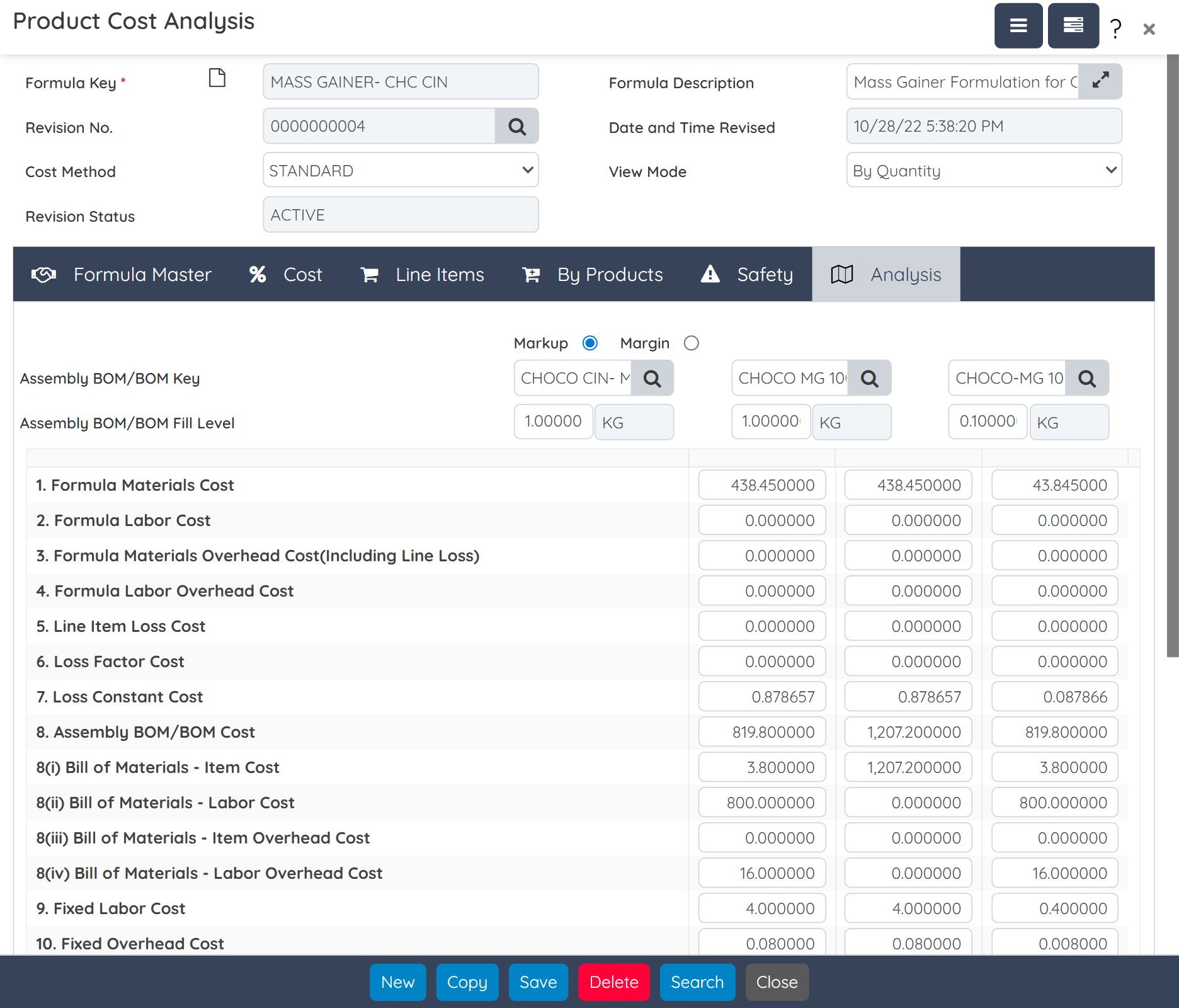
Track real-time batch-wise costs, including raw materials, utilities, labor, and overheads. Calculate actual vs standard cost variances to identify production inefficiencies and increase margins.
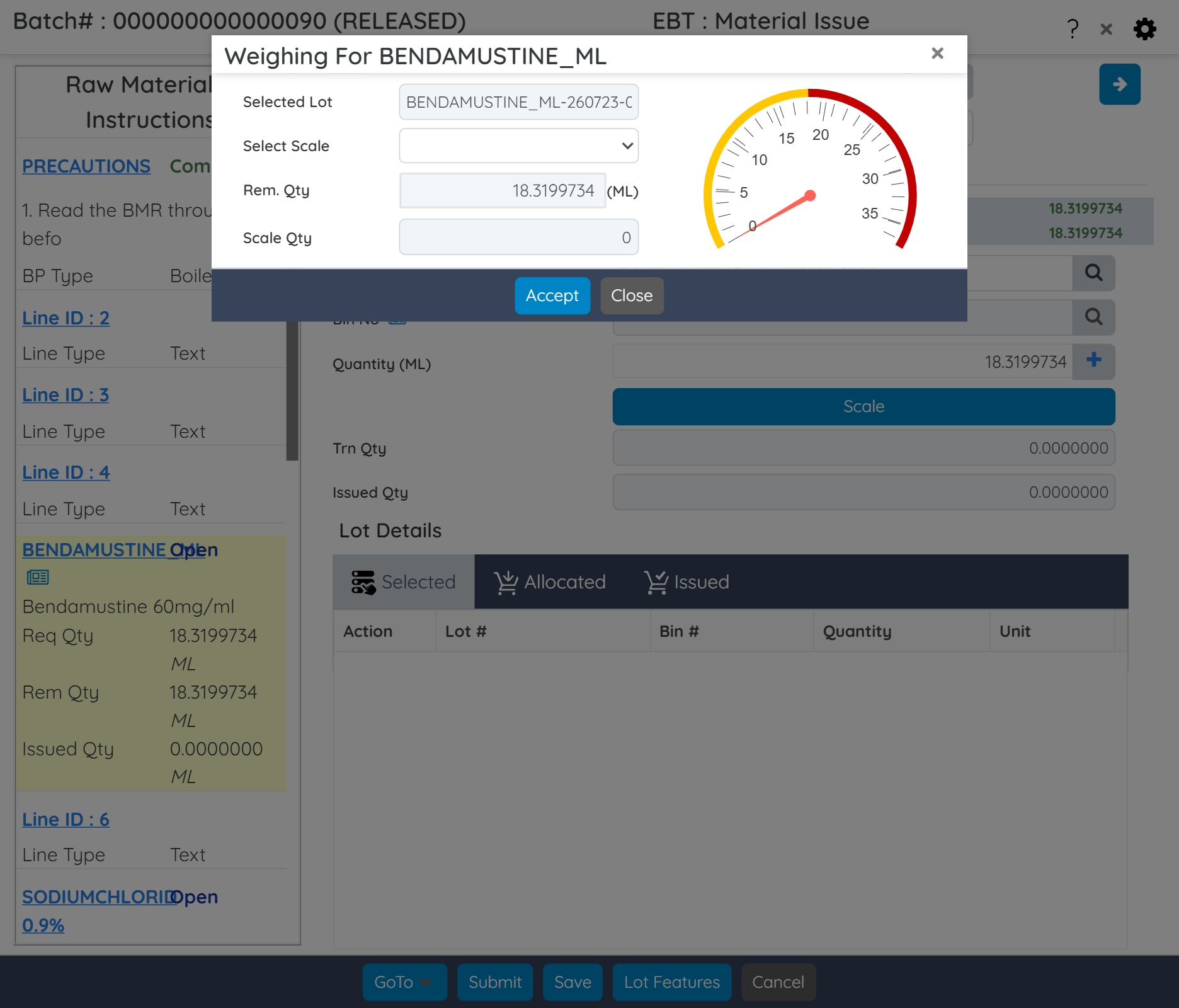
Manage campaign, super batch, and sub-batches across facilities. Digitally capture SOPs and shop-floor activities with integrated weigh sheets and MES tools.
Effectively manage customer-supplied APIs, client-specific formulations, and private labeling with secure portals for order and COA tracking. For own-brand operations, integrate with D2C platforms like Shopify and others.
Automatically generate serial numbers for items and pallets based on customer-specific requirements. Ensure every unit is uniquely identified, traceable, and compliant with global serialization mandates.
Capture and trace critical quality and process data in real time, including medication details, lab timings, temperature readings, and other key process indicators.
Create and manage regulatory documents such as Globally Harmonized System (GHS) labels and Safety Data Sheets (SDS) to meet international compliance standards. BatchMaster ERP provides smooth integration with LISAM to manage these document compliances.













Explore how BatchMaster Pharmaceutical Manufacturing ERP enables manufacturers to eliminate legacy system gaps, maintain audit readiness, and deliver consistent, high-quality products batch after batch.
Success Stories from Leading Pharmaceutical Manufacturers
Integrates with Leading Financial Systems
BatchMaster Pharmaceutical ERP extends the power of your existing financial system by adding industry-specific manufacturing capabilities without disrupting your current operations. Whether you use QuickBooks, SAP Business One, Microsoft Dynamics 365 Business Central, Microsoft Dynamics GP, and Sage 100 & 300, our solution integrates seamlessly to create an end-to-end unified business management platform. The solution is available both on-premises and in the cloud.
Why Manufacturers Choose BatchMaster Pharmaceutical Edition?
BatchMaster ERP for Pharma Industry is a purpose-built ERP solution for manufacturers to grow, stay compliant, and scale efficiently. With industry-best practices embedded, it enables companies to better manage the complexities of pharmaceutical manufacturing while maintaining cost and operational efficiency.


Experience a Smarter Way to Manage Pharmaceutical Manufacturing
Pharmaceutical Manufacturing ERP Resources
BatchMaster Pharmaceutical ERP: FAQs
Generic ERP systems lack the specialized functionality to manage the complexities of pharmaceutical manufacturing. BatchMaster Pharmaceutical Manufacturing ERP is an industry-specific solution that delivers advanced capabilities such as potency management, complete batch traceability, electronic batch records (eBMR), and cGMP-based process controls.
BatchMaster ERP is designed to meet stringent regulatory requirements, including FDA 21 CFR Part 11, cGMP, and other global standards. It offers built-in features such as electronic signatures, secure audit trials, controlled documentation, and end-to-end traceability from raw materials to finished goods.
Our solution enables seamless management of outsourced production by tracking subcontracted batches, overseeing material transfers, enforcing quality checks at every stage. It securely integrates third-party production data into your central records while safeguarding intellectual property.
BatchMaster ERP seamlessly integrates with leading financial and e-commerce platforms, including Shopify, QuickBooks, SAP Business One, Microsoft Dynamics 365, and more. This connectivity creates a unified ecosystem that links production, inventory, sales, and financials for end-to-end operational visibility.
BatchMaster’s MES capabilities bridge the gap between planning and production by managing and monitoring real-time shop floor activities. It guides operators through each step with enforced workflows, integrates with equipment and barcoding systems, and ensures accurate execution. The MES automatically updates inventory, captures production data, generates electronic batch records, and helps deliver consistent, high-quality batches.
















