📚 New Blog Posts
1. KPIs That Drive Enterprise Value for $10–$50M Process Manufacturers (Part 1 of 2)
2. KPIs That Drive Enterprise Value in $10–$50M Process Manufacturers (Part 2 of 2)
3. The Hidden Cost of Choosing the Wrong Manufacturing Software
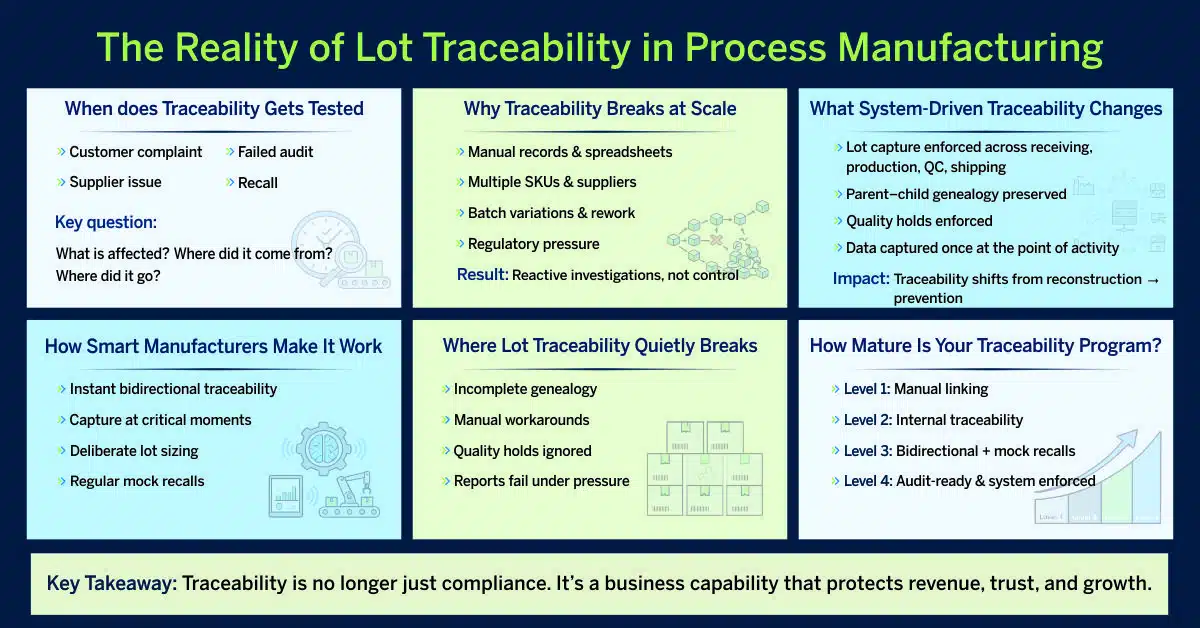
In process manufacturing, lot traceability is rarely questioned when everything is running smoothly. It’s tested when something goes wrong.
- A customer complaint.
- A failed audit.
- A supplier quality issue.
- A recall.
In those moments, manufacturers are expected to answer three questions quickly and confidently:
- What is affected?
- Where did it come from?
- Where did it go?
Recalls grow broader than necessary because affected product can’t be isolated with confidence. Shipments are delayed while teams manually reconstruct records. Customers lose trust when timelines slip or answers change. Audits turn into extended investigations rather than routine validations. Internally, quality, operations, and supply chain teams are pulled into reactive fire drills instead of running the business.
In many cases, the financial and operational impact isn’t driven by the original issue itself. It’s driven by the lack of control over inventory and traceability data.
This is why lot traceability is no longer just a quality or regulatory concern. For growing process manufacturers, it directly affects:
- Recall scope and exposure
- Customer confidence and retention
- Audit readiness and scalability
- The ability to keep inventory moving without risk
Understanding where and why lot traceability breaks is the first step toward preventing these downstream costs
Why Manual and Spreadsheet-Based Traceability Fails at Scale
Before lot traceability was system-driven, most manufacturers didn’t ignore traceability. They managed it with the tools available at the time.
- Paper batch records.
- Spreadsheets maintained by quality or operations teams.
- Receiving logs stored separately from production data.
- QC results filed in binders or shared folders.
- Shipping documents that lived outside manufacturing systems.
Lot numbers existed but they were rarely connected end to end.
A raw material lot might be recorded at receiving, but not consistently validated again at production issue. Batch records showed what was produced, but not always which specific supplier lots were consumed. QC results were reviewed independently, with limited enforcement on whether inventory could be used or shipped. Shipping documents showed what went out, but tracing those shipments back to the exact batch job and ingredient lots required manual reconstruction.
As long as volumes were low and formulations were simple, this approach appeared to work. But it relied heavily on people, memory, and manual coordination.
When issues arose, traceability depended on:
- Experienced employees who “knew how things were done”
- Email chains and phone calls across departments
- Reconciling spreadsheets, paper records, and system reports under time pressure
As operations scaled, this model began to break down.
More SKUs, more suppliers, more batch variations, rework, partial consumption, and tighter regulatory expectations made manual traceability increasingly fragile. Small gaps like missed lot entries, delayed updates, inconsistent records created blind spots that only surfaced during audits, complaints, or recalls.
At that point, traceability shifted from a routine process to a reactive investigation.
Instead of quickly isolating affected inventory, teams were forced to reconstruct events after the fact. The result was broader recalls, delayed responses, internal confusion, and growing uncertainty about what data could actually be trusted.
This isn’t a failure of effort or intent. It’s a limitation of manual, spreadsheet-based traceability in environments where materials transform, combine, split, and move continuously.
And it’s why many manufacturers eventually reach a point where traceability can no longer be managed outside the system and it has to be enforced by it.
What an End-to-End Lot Management System/ERP Changes
An end-to-end lot management system or an ERP doesn’t just make traceability easier to report on. It changes how traceability is executed day to day.
Instead of relying on documentation and reconstruction, traceability becomes a system-enforced process embedded across receiving, production, quality, warehousing, and shipping.
Regulatory shifts such as the FDA’s Food Safety Modernization Act (FSMA) Food Traceability Rule are reinforcing this same reality. Traceability expectations are moving away from post-event reconstruction toward connected, event-level records that can be produced quickly and confidently. For many manufacturers, this has accelerated the shift toward system-enforced traceability embedded directly into receiving, production, quality, and shipping workflows, where traceability is proven through execution and not paperwork.
The most important shift is not visibility but control.
Lot traceability is no longer dependent on people remembering to capture the right information at the right time. It’s built into how work is performed.
In practice, that means:
- Lot control is defined at the item level – Raw materials, intermediates, packaging components, and finished goods are explicitly identified as lot-controlled. Lot number generation rules are standardized, ensuring consistency across receiving and production.
- Lot capture is enforced at receiving and inspection – Materials cannot be received without valid lot identification. Supplier lot numbers are captured and linked to internal lot numbers. QC inspections, COA reviews, and stability tests assign quality status that directly controls whether inventory is available for use.
- Production consumes and produces lots explicitly – During batch execution, raw materials are allocated and consumed by lot number, based on expiration and quality rules. Finished goods and intermediates are assigned new lot numbers at production, preserving parent–child genealogy across blending, splitting, rework, and repack operations.
- Quality status governs movement, not documentation – Holds, releases, and rejections are enforced by the system. Inventory on hold cannot be consumed or shipped, removing reliance on emails, verbal instructions, or manual checks.
- Warehousing and shipping validate lots at execution – Storage, picking, and shipping transactions require lot validation. Finished goods are allocated to customer orders by lot, ensuring what ships matches what was approved.
The result is a traceability model where:
- Data is captured once, at the point of activity
- Every transaction reinforces traceability
- Gaps are prevented instead of discovered later
This is the difference between traceability as a reactive investigation versus a repeatable operational capability, one that continues to hold up as volumes grow, SKUs expand, and regulatory expectations increase.
How Smart Manufacturers Make Lot Traceability Actually Work
Having an end-to-end lot management system in place is necessary but it’s not sufficient on its own.
Manufacturers that consistently contain recalls, pass audits successfully, and keep inventory moving treat lot traceability as an execution discipline, not a reporting feature.
They share a few common practices.
1. They design traceability to work in both directions, instantly
Smart manufacturers don’t think in terms of “forward” or “backward” traceability as separate exercises. They expect to answer either question on demand:
- From a raw material lot → which batches, finished goods, and customers were impacted
- From a finished good lot → which batch job, ingredient lots, and suppliers were involved
This bidirectional view is available if generated in seconds, not hours, and doesn’t require exporting data or manual stitching across reports.
2. They capture traceability at critical moments, not after the fact
Instead of relying on end-of-shift or end-of-day updates, traceability data is captured at the moment of execution which includes receiving, production issue, transformation, QC disposition, and shipping.
This event-based approach ensures that:
- Lot relationships are preserved during blending, splitting, and rework
- Partial consumption doesn’t break genealogy
- Traceability remains accurate even under time pressure
The system becomes the source of truth, not a place to “clean up” data later.
3. They define lots deliberately to balance control and practicality
Lot size isn’t an afterthought.
Mature manufacturers define lot boundaries intentionally, based on:
- Recall containment goals
- Production realities
- Supplier variability
- Quality risk
Overly large lots expand recall exposure. Overly small lots add operational friction. Leading teams find the balance that gives them control without slowing execution.
4. They treat mock recalls as rehearsals, not check-the-box exercises
Mock recalls are run regularly and measured.
Teams know:
- How long it should take to identify affected inventory
- Who is responsible for each step
- Where data gaps still exist
Results are reviewed, documented, and used to improve both system configuration and process discipline.
This is how traceability moves from “we think we can do this” to “we’ve proven we can.”
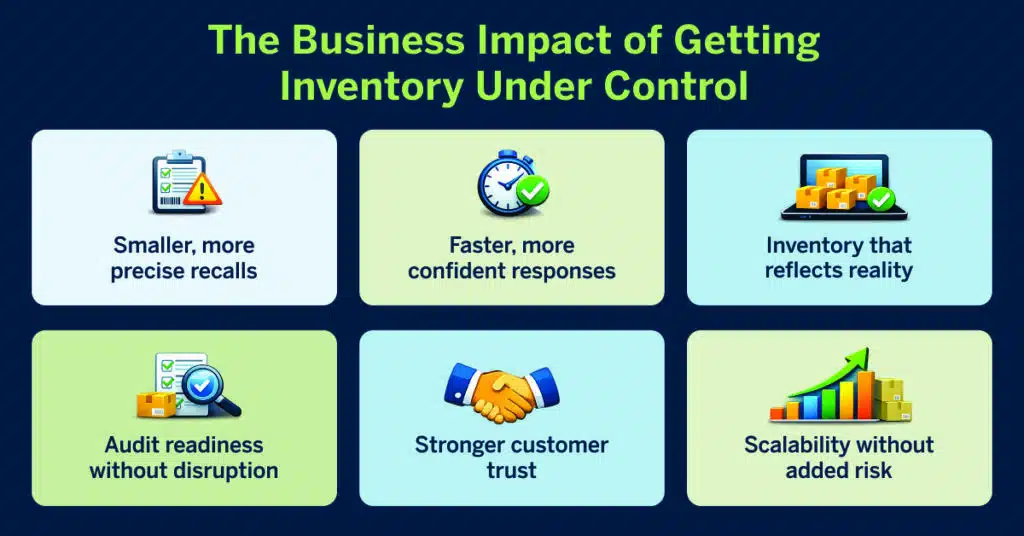
The Business Impact of Getting Inventory Under Control
When lot traceability is system-driven and consistently executed, the benefits extend far beyond regulatory readiness.
Manufacturers gain control over inventory, risk, and response.
The most immediate impact shows up during exceptions. Issues that once triggered widespread disruption become contained, measurable events.
Smaller, more precise recalls
Instead of recalling everything produced during a time window, manufacturers can isolate specific:
- Batches
- Ingredient lots
- Customer shipments
This reduces financial exposure, minimizes customer impact, and shortens recovery time.
Faster, more confident responses
When traceability data is trusted and readily available:
- Investigations move from hours or days to minutes
- Teams spend less time validating data and more time acting
- Communication with customers and regulators is clearer and more consistent
Inventory that reflects reality
System-driven traceability improves inventory accuracy by enforcing:
- Correct lot usage
- Quality-based availability
- Controlled movement
As a result, manufacturers see fewer surprises during cycle counts, audits, and fulfillment.
Inventory becomes something teams rely on and not something they constantly question.
Audit readiness without disruption
Instead of scrambling to assemble documentation, manufacturers can:
- Demonstrate control through execution records
- Trace issues directly through system transactions
- Respond to audit requests without halting operations
Audits become validations and not investigations.
Stronger customer trust
Customers don’t just care that manufacturers can trace products but they care how quickly and clearly answers are delivered when something goes wrong.
Consistent, confident responses build credibility and protect long-term relationships.
Scalability without added risk
As product lines expand, suppliers increase, and volumes grow, manual traceability becomes a constraint.
System-driven traceability allows manufacturers to scale operations without scaling risk, ensuring controls hold up even as complexity increases.
Getting inventory under control isn’t about adding reports or checks. It’s about embedding traceability into daily execution, so when issues arise, the business responds with clarity instead of chaos.
Where Lot Traceability Quietly Breaks in Real Operations
When lot traceability is system-driven, it becomes far more reliable but it’s not immune to breakdowns if execution and configuration aren’t aligned.
In process manufacturing, complexity across materials, transformations, quality controls, and inventory movement can still expose gaps in how traceability is implemented and enforced.
Below are the most common failure points we see when traceability discipline starts to erode in real operations.
1. Lot numbers exist, but genealogy is incomplete
Many manufacturers can see lot numbers on receipts and finished goods, but lack a complete parent–child genealogy across raw materials, intermediates, rework, and packaging.
What this looks like in practice
- Forward trace works; backward trace requires reconstruction
- Rework lots exist, but their origins aren’t clear
- Investigations start in the system and finish in spreadsheets
When genealogy isn’t preserved across transformations, traceability stops being reliable under pressure.
2. Transformations quietly weaken traceability
Blending, splitting, rework, and repack operations are routine in process manufacturing but they’re also where traceability breaks most often.
Common symptoms
- Partial batch consumption creates orphan inventory
- Rework lots aren’t fully linked to original parents
- Lot relationships are overwritten instead of preserved
These gaps usually surface during recalls, not during day-to-day operations.
3. Traceability still depends on manual workarounds
Traceability becomes fragile, if lot accuracy relies on:
- Manual lot entry
- Back-flushing after production
- Optional validation steps
What teams notice
- Lot fields left blank or corrected later
- Month-end inventory adjustments spike
- Physical inventory and system records drift apart
At that point, traceability is only as strong as the least careful transaction.
4. Quality holds are recorded, but not enforced
A lot may be flagged as “on hold” or “pending QC,” yet still remain available for use or shipment.
Why this matters
- QA relies on emails or verbal instructions to stop movement
- “We caught it just in time” becomes a recurring story
- Auditors question whether holds are truly controlled
If a hold doesn’t physically prevent movement, it isn’t a control but a note.
5. Labeling, scanning, and system data drift apart
Traceability often breaks at the edges:
- Receiving docks
- Production floors
- Shipping lanes
Typical signs
- Labels are reprinted manually
- Scans don’t reconcile with reports
- Returns can’t be traced cleanly back to production
These issues rarely appear in reports but they undermine trust in the data.
6. Reporting works until time pressure hits
Many systems can generate traceability reports. The issue is how long it takes and how much interpretation is required.
Symptoms
- Multiple exports and filters
- Different teams produce different answers
- Simple questions turn into multi-hour exercises
During a recall or audit, speed and clarity matter more than report availability.
7. Mock recalls are avoided because they expose gaps
Manufacturers know mock recalls are important but they’re often delayed or minimized.
Why
- They disrupt operations
- They reveal uncomfortable truths
- They require coordination across teams
As a result, the same traceability gaps repeat year after year.
These breakdowns don’t mean traceability systems don’t work. They mean traceability requires intentional design and ongoing discipline.
How Mature Is Your Traceability Program?
Level 1: Lot numbers exist
- But no reliable genealogy; mostly manual linking.
Level 2: Internal traceability works
- Receiving → production → FG is connected; shipping partial.
Level 3: Bidirectional genealogy + recall packets
- Any lot can be traced forward/back quickly; mock recall repeatable.
Level 4: Compliance-ready, audit-ready traceability
- Holds, releases, deviations, and KDE/CTE-style event capture are structured; traceability plan exists (especially in food industry).
Closing Thought
True traceability isn’t achieved through isolated tools or after-the-fact reports.
It requires an end-to-end system that enforces lot discipline across every manufacturing touchpoint, from receiving and quality to production, warehousing, and shipping.
The question for manufacturers isn’t whether traceability exists in theory, but whether their systems and processes are designed to deliver fast, accurate, bi-directional traceability when it’s required.