📚 New Blog Posts
1. This Month in Process Manufacturing – March 2026
2. Why “95% Inventory Accuracy” Still Fails Process Manufacturers
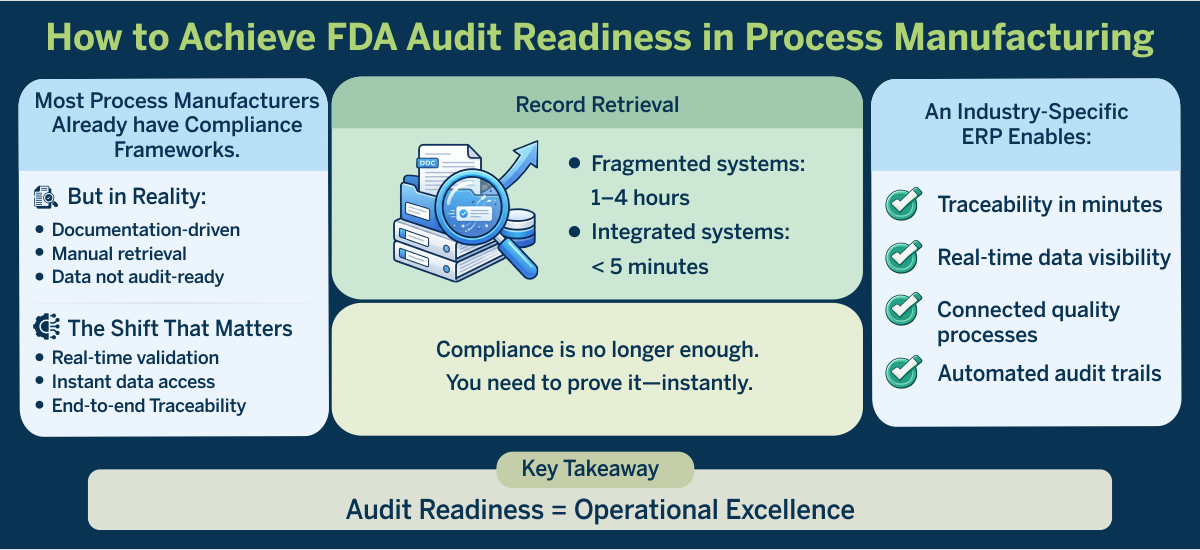
For food & beverage and other regulated manufacturers, FDA audit readiness has fundamentally changed. What was once a documentation-driven exercise is now a test of operational transparency and speed.
Today’s inspections are increasingly unannounced, fast-moving, and data-driven. Investigators no longer accept delays, manual searches, or disconnected records. When they ask for a food safety plan, a supplier verification record, or a traceability report, the expectation is immediate access and not “we’ll get that to you.”
This shift is being driven by tighter regulatory oversight and a clear expectation that manufacturers maintain electronic, organized, and accessible records at all times. In fact, delays in retrieving records are often interpreted as gaps in control, not just inefficiency.
Process manufacturers that still rely on manual processes or fragmented systems are finding that compliance alone is no longer enough. What matters now is the ability to prove compliance in real time.
The Real Problem: Compliance Systems vs. Operational Reality
Most food & beverage and other regulated process manufacturers are not lacking compliance frameworks. They have documented food safety plans, defined SOPs, and required records in place. On paper, they are compliant.
The challenge emerges when these systems are tested in real-world conditions.
In many organizations, compliance lives in documents, while operations run on separate systems and processes. Records are scattered across paper logs, spreadsheets, shared drives, and standalone applications. Data is captured, but not always connected.
The result is a disconnect:
- Records exist, but are difficult to retrieve .
- Processes are defined, but not consistently enforced
- Data is available, but not audit-ready
This gap becomes highly visible during an FDA inspection. Retrieving critical records such as supplier verification or traceability data can take 1–4 hours in fragmented environments, compared to under 5 minutes in integrated systems.
Even seemingly minor delays like checking multiple systems, locating the right person, or searching through files send a strong signal to auditors. What feels like an internal inefficiency is often interpreted as a lack of control or oversight.
At the same time, regulatory scrutiny is increasing. FDA warning letters rose sharply in 2025, reflecting a more aggressive and data-driven enforcement approach.
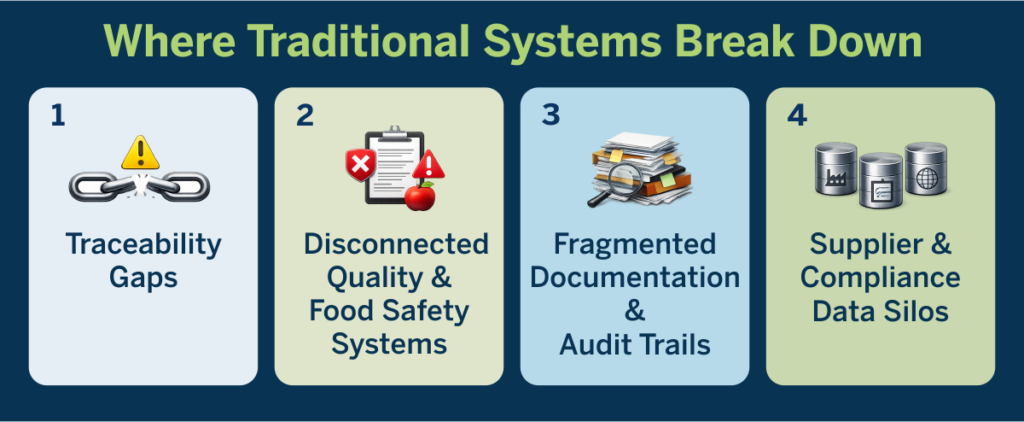
Where Traditional Systems Break Down
Most regulated process manufacturers don’t fail audits because they lack intent or awareness. They fail because the systems supporting compliance are not designed to operate in real time.
As regulatory expectations evolve, the limitations of traditional tools like paper logs, spreadsheets, and disconnected applications become increasingly visible. These systems were built for documentation, not for traceability, validation, or instant retrieval.
The breakdown typically occurs across four critical areas:
1. Traceability Gaps
Traceability is one of the first areas auditors test and one of the most common failure points.
- Lot tracking is often maintained across multiple systems
- Forward and backward traceability requires manual reconciliation
- Mock recalls take hours instead of minutes
With evolving requirements such as FSMA 204, manufacturers are expected to provide traceability data within 24 hours, making manual approaches increasingly unsustainable.
2. Disconnected Quality & Food Safety Systems
Food safety plans exist, but execution is often disconnected from operations.
- Preventive controls are defined but not consistently enforced
- Monitoring records are maintained separately from production data
- Deviations and CAPA processes are tracked manually or outside core systems
According to FDA enforcement trend analyses, many warning letters stem from gaps between documented procedures and actual execution, particularly in preventive controls and sanitation programs.
3. Fragmented Documentation & Audit Trails
Documentation is often spread across:
- Paper records
- Excel sheets
- Shared drives
- Standalone systems
These environments:
- Lack version control
- Provide limited audit trails
- Make it difficult to verify who did what and when
FDA expectations increasingly require secure, time-stamped, and traceable records, especially under regulations aligned with electronic recordkeeping principles.
4. Supplier & Compliance Data Silos
Supplier compliance is another major area of risk.
- Certificates, audits, and approvals are stored in separate systems
- FSVP documentation is incomplete or outdated
- No centralized visibility into supplier status
Given the increasing focus on supply chain accountability, these gaps directly impact audit outcomes.
Where Systems Break: Operational vs. Audit Expectations
| Area | Traditional Approach | Audit Expectation |
| Traceability | Manual lot tracking across systems | Instant forward & backward traceability |
| Record Retrieval | 30 min – several hours | < 5 minutes |
| Quality Monitoring | Paper logs / spreadsheets | Real-time, validated records |
| CAPA & Deviations | Manual tracking | Closed-loop, traceable workflows |
| Supplier Compliance | Disconnected documentation | Centralized, up-to-date records |
| Audit Trail | Limited or none | Time-stamped, tamper-proof records |
What Auditors Actually Test
FDA inspections are not document reviews but they are system validation exercises.
Investigators are trained to assess whether your food safety system is not only documented, but implemented, controlled, and verifiable in real time. Under FSMA’s Preventive Controls framework, inspections focus heavily on execution and not just intent.
In practice, auditors test four critical dimensions:
1. Can You Retrieve Records Immediately?
One of the first things investigators assess is record accessibility.
Under FDA requirements, records must be:
- Readily available
- Accurate and complete
- Provided promptly upon request
In reality, this becomes a live test:
- Food safety plans
- Hazard analysis
- Preventive control records
- Supplier verification documentation
If retrieval takes too long, it raises immediate concerns about control.
From real inspection scenarios:
- Fragmented systems: 1–4 hours retrieval time
- Integrated systems: < 5 minutes
2. Can You Prove That Processes Are Actually Followed?
FDA inspections place strong emphasis on implementation of preventive controls, not just documentation.
Investigators typically:
- Compare SOPs with actual shop floor practices
- Review monitoring logs against defined limits
- Verify whether deviations were properly handled
Common failure pattern:
- SOP exists
- Execution is inconsistent
- Records are incomplete or backfilled
FDA enforcement data consistently shows that many violations stem from gaps between written procedures and real-world execution, particularly in sanitation and preventive controls.
3. Can You Trace a Product Instantly (Forward & Backward)?
Traceability is one of the most critical audit tests.
Investigators may ask you to:
- Trace a finished product back to raw materials
- Identify all customers who received a specific lot
- Demonstrate recall readiness
Under evolving requirements (including FSMA traceability rules), companies must be able to produce traceability data quickly and accurately.
From the report:
- Manual environments: 2–4 hours for traceability exercises
- Digital systems: < 5 minutes
Through this exercise, auditors want to evaluate your ability to respond to a recall scenario in real time
4. Can You Demonstrate Control Over People, Processes, and Suppliers?
FDA inspections also validate broader system control, including:
Training
- Are employees trained on relevant procedures?
- Are training records complete and current?
Supplier Verification (FSVP)
- Are suppliers approved and monitored?
- Are records up to date and accessible?
Corrective Actions (CAPA)
- Were deviations identified and addressed?
- Is there documented follow-through?
These areas often expose system weaknesses:
- Training tracked separately
- Supplier records scattered
- CAPA processes not closed-loop
Through this exercise, auditors want to evaluate whether your organization has end-to-end control across the entire food safety system.
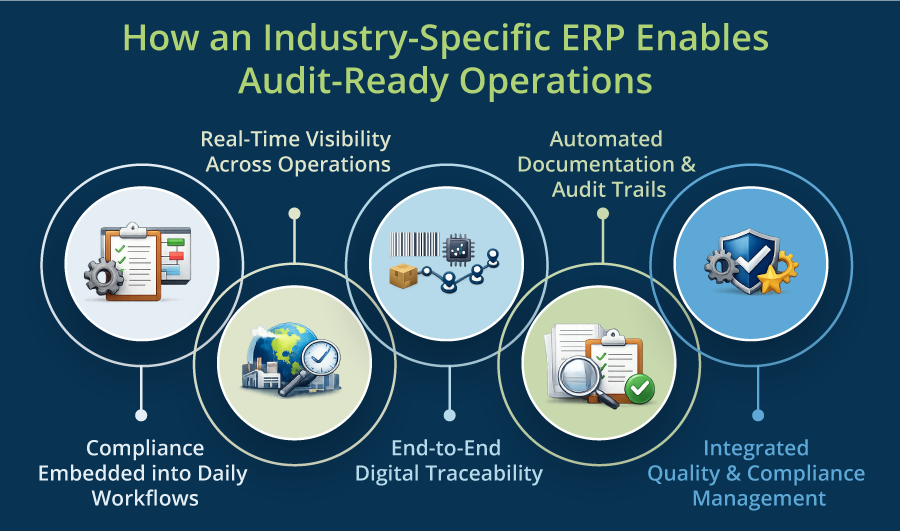
How an Industry-Specific ERP Enables Audit-Ready Operations
Audit readiness today requires a shift from managing compliance separately (paper logs, spreadsheets, and disconnected applications) to embedding compliance directly into operations.
This is where industry-specific ERP systems play a critical role.
Unlike generic systems, an industry-specific ERP designed for food & beverage and other regulated manufacturing connects production, quality, inventory, and compliance into a single, unified environment. The result is not just better recordkeeping but system-enforced compliance, real-time visibility, and end-to-end traceability.
Below are some of the ways that an industry-specific ERP enables audit readiness:
1. Compliance Embedded into Daily Workflows
ERP systems ensure that compliance is not an afterthought.
- Quality checks are triggered during production
- Preventive controls are enforced within workflows
- Deviations automatically initiate corrective actions
This eliminates reliance on manual tracking and ensures that compliance is consistently executed, not just documented.
2. Real-Time Visibility Across Operations
Instead of searching across systems, ERP provides a single source of truth.
- Food safety records are centrally accessible
- Production, quality, and inventory data are connected
- Audit-ready reports can be generated instantly
This aligns directly with FDA expectations for immediate record availability.
3. End-to-End Digital Traceability
ERP systems provide built-in lot traceability across the entire supply chain:
- Raw materials → production → finished goods → distribution
- Forward and backward traceability in minutes
- Faster, more accurate recall execution
This is critical as traceability requirements continue to tighten under FSMA.
4. Automated Documentation & Audit Trails
Every transaction in an ERP system is:
- Time-stamped
- User-tracked
- System-validated
This creates a complete audit trail, eliminating risks associated with:
- Missing records
- Backdated entries
- Version inconsistencies
5. Integrated Quality & Compliance Management
ERP connects food safety plans with execution:
- Monitoring records tied to preventive controls
- CAPA workflows tracked end-to-end
- Supplier compliance managed centrally
This directly addresses one of the most common FDA findings, which is the gap between documented procedures and actual execution.
ERP Audit Readiness Checklist

Not all ERP systems are designed to support FDA compliance. As you evaluate your current system or explore new options, use this checklist to assess whether your organization is truly audit-ready.
1. Industry-Specific Functionality
- Built specifically for process manufacturing (not adapted from discrete)
- Supports regulatory frameworks such as FSMA and GFSI
2. Built-In Traceability
- Lot-level tracking across raw materials, production, and finished goods
- Instant forward and backward traceability
3. Integrated Quality Management
- Preventive controls embedded within workflows
- QC and CAPA processes managed within the system
- No reliance on external tools or manual tracking
4. Real-Time Data Capture
- Mobile or shop floor data entry
- Data captured at the point of activity
- No manual backfilling or delayed entries
5. Centralized Document and Compliance Control
- Version-controlled documents and SOPs
- Centralized access to compliance records
- Instant retrieval during audits
6. Complete Audit Trail and Reporting
- Time-stamped and user-tracked records
- Role-based access and approval workflows
- Audit-ready reports and dashboards
If you find yourself answering “no” or “partially” to multiple items above, it’s a strong indicator that your current systems may not fully support audit-ready operations.
Audit Readiness as a Competitive Advantage
FDA inspections are becoming faster, more data-driven, and less forgiving of delays or inconsistencies. At the same time, regulatory expectations continue to evolve, from FSMA requirements to increasing scrutiny across supply chains and traceability.
In this environment, audit readiness is no longer just about avoiding risk. It has become a reflection of how well a manufacturing operation is controlled, connected, and scalable.
The companies that consistently perform well during inspections are not scrambling to prepare. They operate with systems that provide real-time visibility, built-in compliance, and instant traceability, making every audit a validation of how their business already runs.
For others, the challenge is not a lack of intent or awareness. It is the reliance on systems that require manual effort to prove what should already be visible.
If you’re evaluating how to strengthen audit readiness across your operations, feel free to reach out. We’re happy to share what we’re seeing across process manufacturers and how leading companies are approaching it.