📚 New Blog Posts
1. This Month in Process Manufacturing | April 2026
1. Cloud-Powered Growth: How Cosmetix West Modernized for Global Scale
2. ERP Project Planning & Kickoff: The Foundation for Successful Implementation
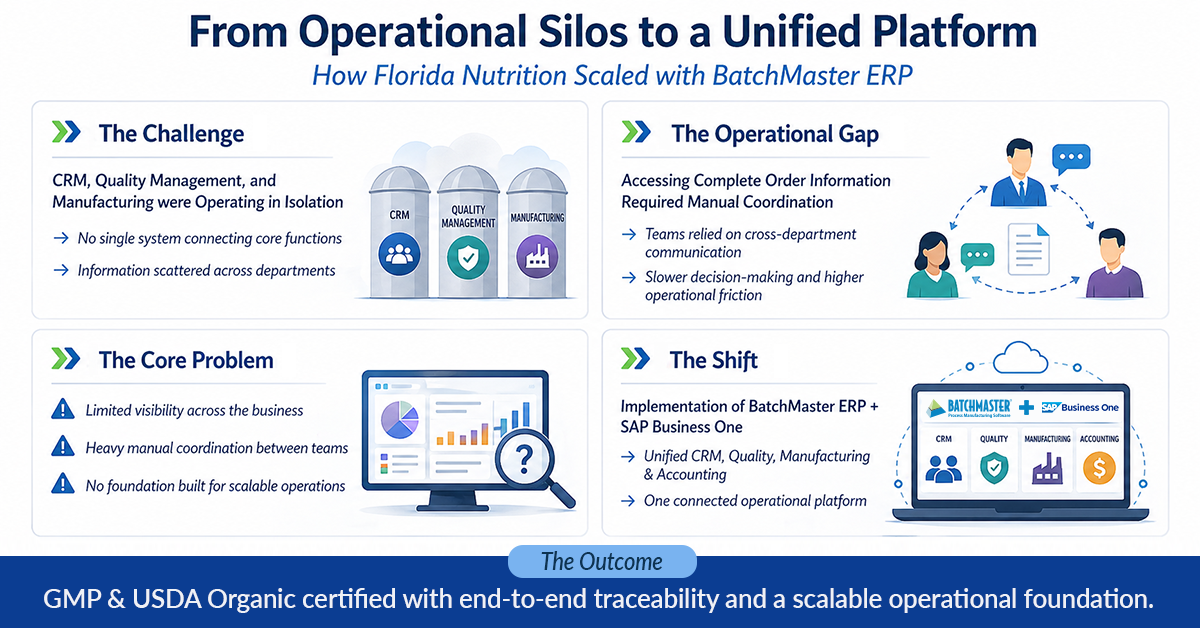
Florida Nutrition is a Tampa-based contract manufacturer of liquid dietary supplements—half-ounce to four-ounce bottles—shipping across the continental United States. A specialist in their category and a recognized leader in what they do, the company’s growth exposed a critical operational gap: too many departments working in isolation, with no single system tying together customer relationships, quality management, and manufacturing. The implementation of BatchMaster ERP with SAP Business One changed that entirely—unifying operations, enabling GMP and USDA Organic certification, and building the scalable foundation the company needed to keep growing.
The Challenge: Too Many Departments, Too Many Systems
Florida Nutrition operates in a focused niche—liquid dietary supplements in a defined size range—but the complexity of managing contract manufacturing is anything but simple. As the company grew, the absence of a unified operational platform created friction across every department.
1. Siloed Departments and Fragmented Information
Key Takeaways:
- Departments operating independently create information gaps that slow every part of the business.
- Without a central system, customer relationship, quality, and manufacturing data cannot be effectively connected.
- Fragmented operations are not a minor inconvenience—they are a strategic ceiling on growth.
What Happened:
Information inaccessible across departments: With each team maintaining its own processes and data, getting a complete picture of any order—from customer requirements through quality checks to manufacturing status—required manual coordination across multiple people and systems. “There were so many different departments doing different things,” says Chris Beekham, COO and Managing Partner. “We needed to find a way to combine everything into one so that all the information was readily available and the company could be run more efficiently.”
Three critical functions operating in isolation: Customer relationship management, quality management, and manufacturing, each operated independently, with no unified platform connecting them. Beekham identified this directly as the core problem to solve: the company needed to “incorporate what we do on the customer relationship side, the quality management side, as well as the manufacturing side.”
2. Compliance and Traceability Without the Right Tools
Key Takeaways:
- In liquid dietary supplement manufacturing, full material traceability is a regulatory requirement—not optional.
- Without the right software, achieving GMP and organic certification is extremely difficult to sustain at scale.
- Traceability infrastructure is a competitive advantage—it unlocks certifications that open new markets.
What Happened:
End-to-end material tracking required by regulation: Liquid dietary supplement manufacturing requires complete documentation at every stage. “The traceability from purchasing the material, receiving the material, to using the material, to testing the material—all has to be recorded and kept for a certain period of time,” Beekham explains. Without an integrated system, meeting this standard reliably and efficiently was a significant operational burden.
Certifications out of reach: In the early days, Florida Nutrition operated as a contract manufacturer without formal certification. Without software capable of systematically tracking all required data, pursuing GMP and USDA Organic certification was not feasible. The compliance documentation demands of these certifications require precisely the kind of integrated, auditable recordkeeping that a purpose-built system provides.
3. A Business That Could Not Scale on Its Existing Infrastructure
Key Takeaways:
- Siloed systems do not just create inefficiency—they actively prevent growth.
- A platform that grows with the business—adding modules as needs evolve—is far more valuable than a static solution.
- The right operational foundation enables faster scaling with less proportional overhead.
What Happened:
Coordination overhead grew with every new order: Each new customer, product line, or compliance requirement added more manual coordination across disconnected departments. The operational model was not designed for the scale Florida Nutrition was targeting.
No path to scalable growth: Without a platform that could scale with the business, Florida Nutrition faced a structural constraint on how quickly it could responsibly grow. What was needed was a system designed not just for their current size, but for where they were going.
The Solution: BatchMaster ERP with SAP Business One
Florida Nutrition selected BatchMaster ERP integrated with SAP Business One, bringing manufacturing, quality management, customer relationship management, and accounting into a single unified platform. The implementation was fast and hands-on: “On day one when BatchMaster came in, they kind of simplified everything, and they were here from start to finish,” says Beekham. “I think it only took a couple weeks—two, three weeks or something—of their time here, and it went pretty quick and was pretty seamless.”
The fit between the platform’s capabilities and Florida Nutrition’s specific need was immediately apparent. “The customization and the ability for BatchMaster and the system to incorporate everything that we needed worked very well with what we wanted,” Beekham explains. “So it was kind of just like a good fit from the beginning.”
The platform gave Florida Nutrition the tools to:
- Unify CRM, quality management, and manufacturing in a single system
- Integrate accounting through SAP Business One, giving any team member a complete cross-functional view
- Maintain complete, audit-ready traceability from material purchase through testing and use
- Add new modules as the business grows, without disrupting existing operations
Key Benefits
| Operational Area | Impact |
| Unified Operations | One person can now drill down into multiple facets of the business—accounting, quality, CRM—from a single system. “Bringing the customer, the CRM part of the program, the customer relationship modules to the table as well brings everything all in one spot,” says Beekham. |
| Compliance & Traceability | Full traceability from material purchasing through receipt, use, and testing is now systematically recorded and retained. This compliance infrastructure is what enabled Florida Nutrition to pursue and achieve GMP and USDA Organic certification. |
| Accounting Integration | SAP Business One brings the accounting function directly into the same platform. Staff can verify payment status, quality results, and customer data without switching systems or waiting for information from other departments. |
| Scalability | New modules can be added as the business grows, without disrupting what is already in place. “As we grow, new modules can be added—new things that they’ve developed and new things that we aren’t currently using. We will be able to scale the company as fast as possible,” Beekham explains. |
| Certifications Achieved | Florida Nutrition has earned GMP and USDA Organic certification since implementing BatchMaster. “Because of the ability of the software and us to track all that stuff, we’ve been able to become certified for GMP and USDA Organic,” says Beekham—certifications that were not in place before the system was implemented. |
Advice for Other Liquid Supplement Contract Manufacturers
Based on Florida Nutrition’s experience, here is what the leadership team would tell other contract manufacturers evaluating their operational infrastructure:
Unification is more valuable than optimization: It is tempting to try to improve individual departmental tools. But the real leverage comes from connecting them. CRM, quality, manufacturing, and accounting working from the same data is a fundamentally different operating model—not just an incremental improvement.
Traceability infrastructure unlocks certification: GMP and USDA Organic certification are competitive differentiators that open doors with customers and in markets. But they are only achievable if your systems can reliably capture, store, and surface the required documentation. Invest in traceability infrastructure early.
Implementation speed matters: Florida Nutrition’s implementation was completed in a few weeks, with BatchMaster on-site throughout. When evaluating vendors, ask about implementation methodology and how hands-on their team will be. A fast, seamless go-live minimizes disruption and accelerates time-to-value.
Choose a platform that grows with you: The ability to add modules as needs evolve means the system remains valuable as the business changes. Avoid solutions that will require replacement when you outgrow them—look for platforms built for where you are going, not just where you are today.
Look for a genuine fit, not a forced one: Beekham describes the BatchMaster relationship as “just like a good fit from the beginning.” That alignment—between platform capability and business need—matters. Bring your actual workflows and requirements to any vendor evaluation, and trust whether the system genuinely supports them.
Conclusion
Florida Nutrition’s experience demonstrates what becomes possible when a manufacturer stops trying to coordinate across disconnected systems and instead builds on a unified operational platform. The company entered the relationship without formal certification and with departments that couldn’t easily share information. Today, they hold GMP and USDA Organic certification, operate with full end-to-end traceability, and have the modular infrastructure to scale as fast as they choose.
“The relationship between SAP and BatchMaster is integral, and we could not see ourselves without that software program,” says Beekham.
For liquid dietary supplement contract manufacturers navigating the same challenges—siloed departments, compliance burdens, and growth ambitions that outpace existing systems—the lesson is clear: the right platform is not a cost center. It is the foundation on which everything else gets built.
Also read the full case study to see how unified systems enabled compliance and growth.
Customer Profile
Company: Florida Nutrition
Industry: Liquid Dietary Supplements
Location: Tampa, Florida
Speaker: Chris Beekham, COO & Managing Partner
Product: BatchMaster ERP with SAP Business One
Key Results
- GMP certification achieved
- USDA Organic certification achieved
- CRM, quality, manufacturing & accounting unified in one platform
- Full end-to-end material traceability
- Fast 2–3 week implementation
- Scalable modular platform for ongoing growth